Effect of endoplasmic reticulum (ER) stress inducer thapsigargin on the expression of extracellular-superoxide dismutase in mouse 3T3-L1 adipocytes
- PMID: 23525536
- PMCID: PMC3593125
- DOI: 10.3164/jcbn.12-46
Effect of endoplasmic reticulum (ER) stress inducer thapsigargin on the expression of extracellular-superoxide dismutase in mouse 3T3-L1 adipocytes
Abstract
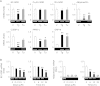
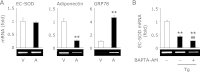
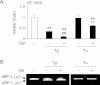
Endoplasmic reticulum stress is related to metabolic disorders, including atherosclerosis and type 2 diabetes. It is known that inflammatory adipocytokines and oxidative stress are increased, while anti-inflammatory adipocytokines such as adiponectin are decreased in adipocytes during above conditions. Extracellular-superoxide dismutase is an anti-inflammatory enzyme that protects cells from oxidative stress. Because plasma extracellular-superoxide dismutase levels in type 2 diabetes patients were inversely related to the body mass index and homeostasis model assessment-insulin resistance index, it is speculated that the regulation of extracellular-superoxide dismutase might lead to the suppression of metabolic disorders. Here, we observed the reduction of extracellular-superoxide dismutase and adiponectin in 3T3-L1 adipocytes treated with thapsigargin, an endoplasmic reticulum stress inducer. Interestingly, tunicamycin, another endoplasmic reticulum stress inducer, did not decrease the expression of extracellular-superoxide dismutase in spite of the induction of glucose regulated protein kinase 78 kDa, an endoplasmic reticulum stress marker. Moreover, eukaryotic translation initiation factor 2α signaling cascade plays a pivotal role in the reduction of extracellular-superoxide dismutase in 3T3-L1 adipocytes during endoplasmic reticulum stress conditions.
Keywords: endoplasmic reticulum stress; eukaryotic translation initiation factor 2α; extracellular-superoxide dismutase; obesity; reactive oxygen species.
Figures

References
-
- Eriksson JW. Metabolic stress in insulin’s target cells leads to ROS accumulation—a hypothetical common pathway causing insulin resistance. FEBS Lett. 2007;581:3734–3742. - PubMed
-
- Ye J, Gao Z, Yin J, He Q. Hypoxia is a potential risk factor for chronic inflammation and adiponectin reduction in adipose tissue of ob/ob and dietary obese mice. Am J Physiol Endocrinol Metab. 2007;293:E1118–E1128. - PubMed
-
- Arita Y, Kihara S, Ouchi N, et al. Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochem Biophys Res Commun. 1999;257:79–83. - PubMed
-
- Ouchi N, Kihara S, Arita Y, et al. Novel modulator for endothelial adhesion molecules: adipocyte-derived plasma protein adiponectin. Circulation. 1999;100:2473–2476. - PubMed
-
- Hosogai N, Fukuhara A, Oshima K, et al. Adipose tissue hypoxia in obesity and its impact on adipocytokine dysregulation. Diabetes. 2007;56:901–911. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

