Protective role of growth hormone against hyperhomocysteinemia-induced glomerular injury
- PMID: 23529346
- PMCID: PMC3654058
- DOI: 10.1007/s00210-013-0848-1
Protective role of growth hormone against hyperhomocysteinemia-induced glomerular injury
Abstract
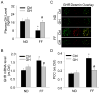
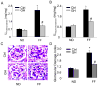
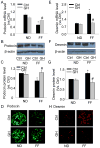
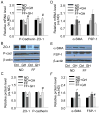
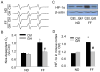
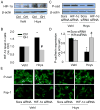
The present study investigated the protective role of growth hormone (GH) against hyperhomocysteinemia (hHcys)-induced activations of reactive oxygen species/hypoxia-inducible factor (HIF)-1α, epithelial-mesenchymal transition (EMT), and consequent glomerular injury. A hHcys model was induced by folate free diet in mice. The urine protein excretion significantly increased while plasma GH levels dramatically decreased in hHcys. Real-time reverse transcription polymerase chain reaction showed that GH receptor (GHR) level increased in the cortex of hHcys mice, which mainly occurred in podocytes as shown by confocal microscopy. Recombinant mouse growth hormone (rmGH) treatment (0.02 mg/kg, once a day for 6 weeks) significantly restored the plasma GH, inhibited GHR upregulation and attenuated proteinuria. Correspondingly, rmGH treatment also blocked hHcys-induced decrease in the expression of podocin, a podocyte slit diaphragm molecule, and inhibited the increases in the expression of desmin, a podocyte injury marker. It was also demonstrated that in hHcys the expression of epithelial markers, p-cadherin and ZO-1, decreased, while the expression of mesenchymal markers, antifibroblast-specific protein 1 (FSP-1) and α-SMA, increased in podocytes, which together suggest the activation of EMT in podocytes. Nicotinamide adenine dinucleotide phosphate oxidase (Nox)-dependent superoxide anion (O2 (.-)) and hypoxia-inducible factor-1α (HIF-1α) level in the hHcys mice cortex was markedly enhanced. These hHcys-induced EMT enhancement and Nox-dependent O2 (.-)/HIF-1α activation were significantly attenuated by rmGH treatment. HIF-1α level increased in Hcys-treated cultured podocytes, which were blocked by rmGH treatment. Meanwhile, homocysteine (Hcys)-induced EMT in cultured podocytes was significantly reversed by HIF-1α siRNA. All these results support the view that GH ameliorates hHcys-induced glomerular injury by reducing Nox-dependent O2 (.-)/HIF-1α signal pathway and EMT.
Conflict of interest statement
None
Figures

Similar articles
-
Epithelial-to-mesenchymal transition in podocytes mediated by activation of NADPH oxidase in hyperhomocysteinemia.Pflugers Arch. 2011 Sep;462(3):455-67. doi: 10.1007/s00424-011-0981-y. Epub 2011 Jun 7. Pflugers Arch. 2011. PMID: 21647593 Free PMC article.
-
Reversal by growth hormone of homocysteine-induced epithelial-to-mesenchymal transition through membrane raft-redox signaling in podocytes.Cell Physiol Biochem. 2011;27(6):691-702. doi: 10.1159/000330078. Epub 2011 Jun 17. Cell Physiol Biochem. 2011. PMID: 21691087 Free PMC article.
-
Nod-like receptor protein 3 (NLRP3) inflammasome activation and podocyte injury via thioredoxin-interacting protein (TXNIP) during hyperhomocysteinemia.J Biol Chem. 2014 Sep 26;289(39):27159-27168. doi: 10.1074/jbc.M114.567537. Epub 2014 Aug 19. J Biol Chem. 2014. PMID: 25138219 Free PMC article.
-
Detrimental effects of hypoxia on glomerular podocytes.J Physiol Biochem. 2021 May;77(2):193-203. doi: 10.1007/s13105-021-00788-y. Epub 2021 Apr 9. J Physiol Biochem. 2021. PMID: 33835424 Review.
-
Hyperhomocysteinemia: Metabolic Role and Animal Studies with a Focus on Cognitive Performance and Decline-A Review.Biomolecules. 2021 Oct 19;11(10):1546. doi: 10.3390/biom11101546. Biomolecules. 2021. PMID: 34680179 Free PMC article. Review.
Cited by
-
Catalpol inhibits HHcy-induced EndMT in endothelial cells by modulating ROS/NF-κB signaling.BMC Cardiovasc Disord. 2024 Aug 16;24(1):431. doi: 10.1186/s12872-024-04046-z. BMC Cardiovasc Disord. 2024. PMID: 39148029 Free PMC article.
-
Omega-3FAs Can Inhibit the Inflammation and Insulin Resistance of Adipose Tissue Caused by HHcy Induced Lipids Profile Changing in Mice.Front Physiol. 2021 Feb 12;12:628122. doi: 10.3389/fphys.2021.628122. eCollection 2021. Front Physiol. 2021. PMID: 33643070 Free PMC article.
-
Protective Action of Anandamide and Its COX-2 Metabolite against l-Homocysteine-Induced NLRP3 Inflammasome Activation and Injury in Podocytes.J Pharmacol Exp Ther. 2016 Jul;358(1):61-70. doi: 10.1124/jpet.116.233239. Epub 2016 May 11. J Pharmacol Exp Ther. 2016. PMID: 27189966 Free PMC article.
-
(Pro)renin receptor contributes to renal mitochondria dysfunction, apoptosis and fibrosis in diabetic mice.Sci Rep. 2019 Aug 12;9(1):11667. doi: 10.1038/s41598-019-47055-1. Sci Rep. 2019. PMID: 31406124 Free PMC article.
-
(Pro)renin receptor regulates autophagy and apoptosis in podocytes exposed to high glucose.Am J Physiol Endocrinol Metab. 2015 Aug 1;309(3):E302-10. doi: 10.1152/ajpendo.00603.2014. Epub 2015 Jun 16. Am J Physiol Endocrinol Metab. 2015. PMID: 26081285 Free PMC article.
References
-
- Arnold RE, Weigent DA. The inhibition of superoxide production in EL4 lymphoma cells overexpressing growth hormone. Immunopharmacol Immunotoxicol. 2003;25:159–177. - PubMed
-
- Chen YF, Li PL, Zou AP. Effect of hyperhomocysteinemia on plasma or tissue adenosine levels and renal function. Circulation. 2002;106:1275–1281. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

