Normal dendritic cell mobilization to lymph nodes under conditions of severe lymphatic hypoplasia
- PMID: 23530147
- PMCID: PMC3634097
- DOI: 10.4049/jimmunol.1202600
Normal dendritic cell mobilization to lymph nodes under conditions of severe lymphatic hypoplasia
Abstract
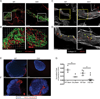
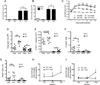
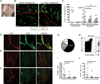
To address the requirement for lymphatic capillaries in dendritic cell (DC) mobilization from skin to lymph nodes (LNs), we used mice bearing one inactivated allele of vascular endothelial growth factor receptor 3 (VEGFR3) where skin lymphatic capillaries are reported absent. Unexpectedly, DC mobilization from the back skin to draining LNs was similar in magnitude, and kinetics to control mice and humoral immunity appeared intact. By contrast, DC migration from body extremities, including ear and forepaws, was ablated. An evaluation in different regions of skin revealed rare patches of lymphatic capillaries only in body trunk areas where migration was intact. That is, whereas the ear skin was totally devoid of lymphatic capillaries, residual capillaries in the back skin were present though retained only at ∼10% normal density. This reduction in density markedly reduced the clearance of soluble tracers, indicating that normal cell migration was spared under conditions when lymphatic transport function was poor. Residual lymphatic capillaries expressed slightly higher levels of CCL21 and migration of skin DCs to LNs remained dependent on CCR7 in Chy mice. DC migration from the ear could be rescued by the introduction of a limited number of lymphatic capillaries through skin transplantation. Thus, the development of lymphatic capillaries in the skin of body extremities was more severely impacted by a mutant copy of VEGFR3 than trunk skin, but lymphatic transport function was markedly reduced throughout the skin, demonstrating that even under conditions when a marked loss in lymphatic capillary density reduces lymph transport, DC migration from skin to LNs remains normal.
Figures




References
-
- Randolph GJ, Angeli V, Swartz MA. Dendritic-cell trafficking to lymph nodes through lymphatic vessels. Nat Rev Immunol. 2005;5:617–628. - PubMed
-
- Schmid-Schonbein GW. Microlymphatics and lymph flow. Physiol Rev. 1990;70:987–1028. - PubMed
-
- Muthuchamy M, Zawieja D. Molecular regulation of lymphatic contractility. Ann N Y Acad Sci. 2008;1131:89–99. - PubMed
-
- Rockson SG. Causes and consequences of lymphatic disease. Ann N Y Acad Sci. 2010;1207(Suppl 1):E2–E6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

