Economic evaluation of 5-grass pollen tablets versus placebo in the treatment of allergic rhinitis in adults
- PMID: 23532801
- PMCID: PMC3634979
- DOI: 10.1007/s40261-013-0067-z
Economic evaluation of 5-grass pollen tablets versus placebo in the treatment of allergic rhinitis in adults
Abstract
Background: Allergen immunotherapy (AIT) is aimed at modifying the immune response to a causative allergen, thereby reducing clinical symptoms and symptomatic medication intake and improving quality of life. Long-term AIT research has led to the development of 5-grass pollen tablets, currently indicated for the treatment of grass pollen-induced allergic rhinitis (AR).
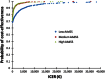
Methods: A post-hoc analysis was conducted using the Average Adjusted Symptom Score (AAdSS) to compare the effect of treatment of AR with 5-grass pollen tablets versus placebo treatment. Using the results of the VO34.04 and VO53.06 trials and economic data, cost-effectiveness analysis of 5-grass pollen tablet treatment was performed from the Italian third-party payer perspective with cost data derived from a study of 2008 updated to 2011. Also a societal perspective was considered by using the costs related to the losses of productivity by following the human capital approach. Using the results of the analysis, the estimated receiver-operating characteristic curve was plotted to evaluate medication effectiveness in terms of quality-adjusted life years (QALYs) and a decision tree constructed to model the possible outcomes and costs for adults and paediatric patients with a low, medium, and high AAdSS. Finally, probabilistic sensitivity analysis was conducted to test the robustness of the results as well as their consistency at an assumed cost-effectiveness threshold of € 30,000/QALY.
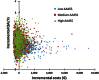
Results: The results indicate that compared to the placebo, the 5-grass pollen tablet treatment provides a benefit of 0.127 QALYs in medium AAdSS patients and of 0.143 QALYs in high AAdSS patients. The 5-grass pollen tablet treatment was found to cost € 1,024/QALY for patients with a medium AAdSS and € 1,035/QALY for patients with a high AAdSS. Of all the simulations performed in the probabilistic sensitivity analysis, 99 % indicated that the incremental cost-effectiveness ratio of the 5-grass pollen tablet treatment was below the threshold of € 30,000/QALY in patients with medium and high AAdSS, whereas it was found to be dominated in 67 % of simulations related to patients with low AAdSS.
Conclusion: The 5-grass pollen tablet is a cost-effective treatment for adult AR patients with a medium or high AAdSS. This finding should be carefully considered when deciding the management strategy for these patients.
Figures

References
-
- Malone DC, Lawson KA, Smith DH, et al. A cost of illness study of allergic rhinitis in the United States. J Allergy Clin Immunol. 1997;99:22–27. - PubMed
-
- Mackowiak JI. The health and economic impact of rhinitis. Am J Manag Care. 1997;3:S8–S18.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

