Ferumoxytol: a new, clinically applicable label for stem-cell tracking in arthritic joints with MRI
- PMID: 23534832
- PMCID: PMC3816126
- DOI: 10.2217/nnm.12.198
Ferumoxytol: a new, clinically applicable label for stem-cell tracking in arthritic joints with MRI
Abstract
Aim: To develop a clinically applicable MRI technique for tracking stem cells in matrix-associated stem-cell implants, using the US FDA-approved iron supplement ferumoxytol.
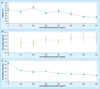
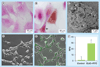
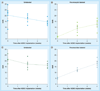
Materials & methods: Ferumoxytol-labeling of adipose-derived stem cells (ADSCs) was optimized in vitro. A total of 11 rats with osteochondral defects of both femurs were implanted with ferumoxytol- or ferumoxides-labeled or unlabeled ADSCs, and underwent MRI up to 4 weeks post matrix-associated stem-cell implant. The signal-to-noise ratio of different matrix-associated stem-cell implant was compared with t-tests and correlated with histopathology.
Results: An incubation concentration of 500 µg iron/ml ferumoxytol and 10 µg/ml protamine sulfate led to significant cellular iron uptake, T2 signal effects and unimpaired ADSC viability. In vivo, ferumoxytol- and ferumoxides-labeled ADSCs demonstrated significantly lower signal-to-noise ratio values compared with unlabeled controls (p < 0.01). Histopathology confirmed engraftment of labeled ADSCs, with slow dilution of the iron label over time.
Conclusion: Ferumoxytol can be used for in vivo tracking of stem cells with MRI.
Conflict of interest statement
The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
Figures






Similar articles
-
Intravenous ferumoxytol allows noninvasive MR imaging monitoring of macrophage migration into stem cell transplants.Radiology. 2012 Sep;264(3):803-11. doi: 10.1148/radiol.12112393. Epub 2012 Jul 19. Radiology. 2012. PMID: 22820731 Free PMC article.
-
Iron administration before stem cell harvest enables MR imaging tracking after transplantation.Radiology. 2013 Oct;269(1):186-97. doi: 10.1148/radiol.13130858. Epub 2013 Jul 12. Radiology. 2013. PMID: 23850832 Free PMC article.
-
MegaPro, a clinically translatable nanoparticle for in vivo tracking of stem cell implants in pig cartilage defects.Theranostics. 2023 Apr 29;13(8):2710-2720. doi: 10.7150/thno.82620. eCollection 2023. Theranostics. 2023. PMID: 37215574 Free PMC article.
-
Magnetic resonance imaging of stem cell-macrophage interactions with ferumoxytol and ferumoxytol-derived nanoparticles.Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2019 Jul;11(4):e1552. doi: 10.1002/wnan.1552. Epub 2019 Feb 7. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2019. PMID: 30734542 Free PMC article. Review.
-
Superparamagnetic iron oxide nanoparticles as MRI contrast agents for non-invasive stem cell labeling and tracking.Theranostics. 2013 Jul 31;3(8):595-615. doi: 10.7150/thno.5366. eCollection 2013. Theranostics. 2013. PMID: 23946825 Free PMC article. Review.
Cited by
-
Iron-based superparamagnetic nanoparticle contrast agents for MRI of infection and inflammation.AJR Am J Roentgenol. 2015 Mar;204(3):W302-13. doi: 10.2214/AJR.14.12733. AJR Am J Roentgenol. 2015. PMID: 25714316 Free PMC article. Review.
-
Fluorine-19 Labeling of Stromal Vascular Fraction Cells for Clinical Imaging Applications.Stem Cells Transl Med. 2015 Dec;4(12):1472-81. doi: 10.5966/sctm.2015-0113. Epub 2015 Oct 28. Stem Cells Transl Med. 2015. PMID: 26511652 Free PMC article. Clinical Trial.
-
Exceedingly small iron oxide nanoparticles as positive MRI contrast agents.Proc Natl Acad Sci U S A. 2017 Feb 28;114(9):2325-2330. doi: 10.1073/pnas.1620145114. Epub 2017 Feb 13. Proc Natl Acad Sci U S A. 2017. PMID: 28193901 Free PMC article.
-
Dendritic cell activation by iron oxide nanoparticles depends on the extracellular environment.Nanoscale Adv. 2024 Nov 12;7(1):209-218. doi: 10.1039/d4na00561a. eCollection 2024 Dec 17. Nanoscale Adv. 2024. PMID: 39569333 Free PMC article.
-
Receptor-Mediated SPION Labeling of CD4+ T Cells for Longitudinal MRI Tracking of Distribution Following Systemic Injection in Mouse.Nanomaterials (Basel). 2025 Jul 10;15(14):1068. doi: 10.3390/nano15141068. Nanomaterials (Basel). 2025. PMID: 40711188 Free PMC article.
References
-
- Centers for Disease Control and Prevention. Prevalence of doctor-diagnosed arthritis and arthritis-attributable activity limitation – United States, 2007–2009. MMWR Morb. Mortal. Wkly Rep. 2010;59(39):1261–1265. - PubMed
-
- Centers for Disease Control and Prevention. Arthritis. Meeting the Challenge at a Glance 2011. GA, USA: National Center for Chronic Disease Prevention and Health Promotion; 2011.
-
- Hunziker EB. Articular cartilage repair: problems and perspectives. Biorheology. 2000;37(1–2):163–164. - PubMed
-
- Kinner B, Capito RM, Spector M. Regeneration of articular cartilage. Adv. Biochem. Eng. Biotechnol. 2005;94:91–123. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
