Estradiol differentially regulates calreticulin: a potential link with abnormal T cell function in systemic lupus erythematosus?
- PMID: 23535532
- PMCID: PMC4072130
- DOI: 10.1177/0961203313482742
Estradiol differentially regulates calreticulin: a potential link with abnormal T cell function in systemic lupus erythematosus?
Abstract
Objective: Systemic lupus erythematosus (SLE) is an autoimmune disease that affects women nine times more often than men. The present study investigates estradiol-dependent control of the calcium-buffering protein, calreticulin, to gain further insight into the molecular basis of abnormal T cell signaling in SLE T cells.
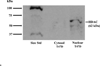
Methods: T cells were purified from blood samples obtained from healthy females and SLE patients. Calreticulin expression was quantified by real-time polymerase chain amplification. Calreticulin and estrogen receptor-α were co-precipitated and analyzed by Western blotting to determine if the proteins associate in T cells.
Results: Calreticulin expression increased (p = 0.034) in activated control T cells, while estradiol decreased (p = 0.044) calreticulin in resting T cells. Calreticulin expression decreased in activated SLE T cell samples and increased in approximately 50% of resting T cell samples. Plasma estradiol was similar (p > 0.05) among SLE patients and control volunteers. Estrogen receptor-α and calreticulin co-precipitated from nuclear and cytoplasmic T cell compartments.
Conclusions: The results indicate that estradiol tightly regulates calreticulin expression in normal human T cells, and the dynamics are different between activated and resting T cells. The absence of this tight regulation in SLE T cells could contribute to abnormal T cell function.
Keywords: SLE; calreticulin; estradiol; estrogen receptor-α; human T cells.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Figures












Similar articles
-
Gender Bias in Human Systemic Lupus Erythematosus: A Problem of Steroid Receptor Action?Front Immunol. 2018 Mar 28;9:611. doi: 10.3389/fimmu.2018.00611. eCollection 2018. Front Immunol. 2018. PMID: 29643853 Free PMC article. Clinical Trial.
-
Differential expression of estrogen receptors in women with systemic lupus erythematosus.J Rheumatol. 2006 Jun;33(6):1093-101. J Rheumatol. 2006. PMID: 16755656
-
Estrogen receptor α regulates tripartite motif-containing protein 21 expression, contributing to dysregulated cytokine production in systemic lupus erythematosus.Arthritis Rheumatol. 2014 Jan;66(1):163-72. doi: 10.1002/art.38187. Arthritis Rheumatol. 2014. PMID: 24449583
-
Attempt to correct abnormal signal transduction in T lymphocytes from systemic lupus erythematosus patients.Autoimmun Rev. 2006 Feb;5(2):143-4. doi: 10.1016/j.autrev.2005.09.004. Epub 2005 Sep 19. Autoimmun Rev. 2006. PMID: 16431347 Review.
-
T cell abnormalities in systemic lupus erythematosus.Autoimmunity. 2005 Aug;38(5):339-46. doi: 10.1080/08916930500123983. Autoimmunity. 2005. PMID: 16227148 Review.
Cited by
-
Sex Hormones in Acquired Immunity and Autoimmune Disease.Front Immunol. 2018 Oct 4;9:2279. doi: 10.3389/fimmu.2018.02279. eCollection 2018. Front Immunol. 2018. PMID: 30337927 Free PMC article. Review.
-
Role of Plasma Calreticulin in the Prediction of Severity in Septic Patients.Dis Markers. 2019 Sep 12;2019:8792640. doi: 10.1155/2019/8792640. eCollection 2019. Dis Markers. 2019. PMID: 31612071 Free PMC article.
References
-
- Cervera R, Khamashta MA, Font J, et al. The European Working Party on Systemic Lupus Erythematosus. Vol. 72. Medicine (Baltimore); 1993. Systemic lupus erythematosus: clinical and immunologic patterns of disease expression in a cohort of 1,000 patients; pp. 113–124. - PubMed
-
- Tenbrock K, Juang YT, Kyttaris VC, et al. Altered signal transduction in SLE T cells. Rheumatology. 2007;46:1525–1530. - PubMed
-
- Kammer GM. Deficient protein kinase A in systemic lupus erythematosus: a disorder of T lymphocyte signal transduction. Ann NY Acad Sci. 2002;968:96–105. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

