Effects of varenicline and bupropion sustained-release use plus intensive smoking cessation counseling on prolonged abstinence from smoking and on depression, negative affect, and other symptoms of nicotine withdrawal
- PMID: 23536105
- PMCID: PMC4128024
- DOI: 10.1001/jamapsychiatry.2013.678
Effects of varenicline and bupropion sustained-release use plus intensive smoking cessation counseling on prolonged abstinence from smoking and on depression, negative affect, and other symptoms of nicotine withdrawal
Abstract
Importance: Given the actions of varenicline tartrate and bupropion hydrochloride sustained-release (SR) on neurobiological targets related to affect and reward, it is thought that the modulation of nicotine withdrawal symptoms may contribute to their effectiveness.
Objective: To assess the relative efficacy of varenicline and bupropion SR plus intensive counseling on smoking cessation and emotional functioning.
Design and setting: Placebo-controlled randomized clinical trial at a university medical center.
Participants: In total, 294 community volunteers who wanted to quit smoking.
Interventions: Twelve weeks of varenicline, bupropion SR, or placebo plus intensive smoking cessation counseling (10 sessions, for a total of approximately 240 minutes of counseling).
Main outcome measures: Prolonged abstinence from smoking and weekly measures of depression, negative affect, and other symptoms of nicotine withdrawal.
Results: Significant differences were found in abstinence at the end of treatment and through the 3-month postquit follow-up visit, favoring both active medications compared with placebo. At the 6-month postquit follow-up visit, only the varenicline vs placebo comparison remained significant. Varenicline use was also associated with a generalized suppression of depression and reduced smoking reward compared with the other treatments, while both active medications improved concentration, reduced craving, and decreased negative affect and sadness compared with placebo, while having little effect (increase or decrease) on anxiety and anger. No differences were noted in self-reported rates of neuropsychiatric adverse events.
Conclusions and relevance: In a community sample, varenicline exerts a robust and favorable effect on smoking cessation relative to placebo and may have a favorable (suppressive) effect on symptoms of depression and other affective measures, with no clear unfavorable effect on neuropsychiatric adverse events.
Trial registration: clinicaltrials.gov Identifier: NCT00507728.
Conflict of interest statement
The other authors declare no conflict of interest.
Figures

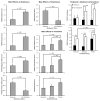
Comment in
-
Varenicline may reduce negative effect while aiding smoking cessation.Evid Based Med. 2014 Feb;19(1):23. doi: 10.1136/eb-2013-101435. Epub 2013 Aug 29. Evid Based Med. 2014. PMID: 23990527 No abstract available.
References
-
- Centers for Disease Control and Prevention. Quitting Smoking Among Adults - United States, 2001–2010. Morbidity and Mortality Weekly Report. 2011;60:1513–1519. - PubMed
-
- Fiore MC, Jaen CR, Baker TB, Bailey WC, Benowitz NL, Curry SJ, Dorfman SF, Froelicher ES, Goldstein MG, Healton CG, Henderson PN, Heyman RB, Koh HK, Kottke TE, Lando HA, Mecklenburg RE, Mullen PD, Orleans CT, Robinson LA, Stitzer ML, Tommasello AC, Villejo L, Wewers ME. Treating Tobacco Use and Dependence: 2008 Update, Clinical Practice Guideline. Rockville, MD: U. S. Department of Health and Human Services. Public Health Service; 2008.
-
- Cahill K, Stead LF, Lancaster T. Nicotine receptor partial agonists for smoking cessation. Cochrane Database of Systematic Reviews. 2011;2:1–87.
-
- Hughes JR, Stead LF, Lancaster T. Antidepressants for smoking cessation. Cochrane Database of Systematic Reviews. 2007;2007:1, CD000031. - PubMed
-
- Nides M, Glover ED, Reus VI, Christen AG, Make BJ, Billing CB, Jr, Williams KE. Varenicline versus bupropion SR or placebo for smoking cessation: a pooled analysis. American Journal of Health Behavior. 2008;32:664–675. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

