Selenium toxicity from a misformulated dietary supplement, adverse health effects, and the temporal response in the nail biologic monitor
- PMID: 23538937
- PMCID: PMC3705333
- DOI: 10.3390/nu5041024
Selenium toxicity from a misformulated dietary supplement, adverse health effects, and the temporal response in the nail biologic monitor
Abstract
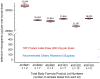
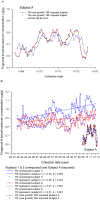
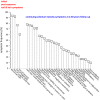
Use of dietary supplements in the U.S. has increased steadily over the last 25 years. While misformulation is uncommon, the consequences can be serious. A March 2008 voluntary market recall removed supplement products responsible for the most serious selenium toxicity outbreak that has occurred in the U.S. We quantified selenium concentrations in the misformulated supplement products, measured the temporal response in the nail biologic monitor, and associated exposure to self-reported selenosis symptoms. Subjects recruited through state health departments and referrals provided samples of the misformulated supplement products, exposure information, monthly toenail and or fingernail clippings or onycholysitic nail fragments, and listed their newly onset adverse health effects attributed to selenium toxicity. Ninety-seven subjects enrolled and submitted at least one test sample. Peak selenium concentrations (up to 18.3 and 44.1 μg/g for toenails and fingernails, respectively) were measured. Multiple samples (52 total) of all six recalled supplement lots were analyzed ranging from 22,300 to 32,200 μg selenium per daily dose. Average consumption was 30.9 ± 13.9 doses; 73 subjects provided follow-up data on selenosis symptoms at 2.50 ± 0.14 years. Nail samples accurately reflect exposure in this selenium toxicity outbreak, which resulted in long-term/permanent adverse health effects.
Figures



References
-
- Institute of Medicine, The National Academies. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium and Carotenoids. National Academy Press; Washington, DC, USA: 2000. - PubMed
-
- Xia Y., Hill K.E., Byrne D.W., Xu J., Burk R.F. Effectiveness of selenium supplements in a low-selenium area of China. Am. J. Clin. Nutr. 2005;81:829–834. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

