Selection of distinct populations of dentate granule cells in response to inputs as a mechanism for pattern separation in mice
- PMID: 23538967
- PMCID: PMC3602954
- DOI: 10.7554/eLife.00312
Selection of distinct populations of dentate granule cells in response to inputs as a mechanism for pattern separation in mice
Abstract
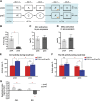
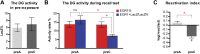
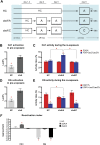
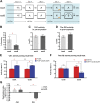
The hippocampus is critical for episodic memory and computational studies have predicted specific functions for each hippocampal subregion. Particularly, the dentate gyrus (DG) is hypothesized to perform pattern separation by forming distinct representations of similar inputs. How pattern separation is achieved by the DG remains largely unclear. By examining neuronal activities at a population level, we revealed that, unlike CA1 neuron populations, dentate granule cell (DGC) ensembles activated by learning were not preferentially reactivated by memory recall. Moreover, when mice encountered an environment to which they had not been previously exposed, a novel DGC population-rather than the previously activated DGC ensembles that responded to past events-was selected to represent the new environmental inputs. This selection of a novel responsive DGC population could be triggered by small changes in environmental inputs. Therefore, selecting distinct DGC populations to represent similar but not identical inputs is a mechanism for pattern separation. DOI:http://dx.doi.org/10.7554/eLife.00312.001.
Keywords: CA1; Mouse; dentate gyrus; hippocampus; learning and memory; pattern separation.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures
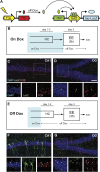

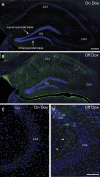
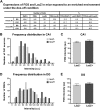
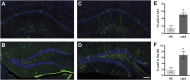






Comment in
-
Learning and memory: remembrance of things similar..Nat Rev Neurosci. 2013 May;14(5):306-7. doi: 10.1038/nrn3487. Epub 2013 Apr 5. Nat Rev Neurosci. 2013. PMID: 23558970 No abstract available.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

