Effects of prostaglandins and COX-inhibiting drugs on skeletal muscle adaptations to exercise
- PMID: 23539318
- PMCID: PMC3764617
- DOI: 10.1152/japplphysiol.00061.2013
Effects of prostaglandins and COX-inhibiting drugs on skeletal muscle adaptations to exercise
Abstract
It has been ∼40 yr since the discovery that PGs are produced by exercising skeletal muscle and since the discovery that inhibition of PG synthesis is the mechanism of action of what are now known as cyclooxygenase (COX)-inhibiting drugs. Since that time, it has been established that PGs are made during and after aerobic and resistance exercise and have a potent paracrine and autocrine effect on muscle metabolism. Consequently, it has also been determined that orally consumed doses of COX inhibitors can profoundly influence muscle PG synthesis, muscle protein metabolism, and numerous other cellular processes that regulate muscle adaptations to exercise loading. Although data from acute human exercise studies, as well as animal and cell-culture data, would predict that regular consumption of a COX inhibitor during exercise training would dampen the typical muscle adaptations, the chronic data do not support this conjecture. From the studies in young and older individuals, lasting from 1.5 to 4 mo, no interfering effects of COX inhibitors on muscle adaptations to resistance-exercise training have been noted. In fact, in older individuals, a substantial enhancement of muscle mass and strength has been observed. The collective findings of the PG/COX-pathway regulation of skeletal muscle responses and adaptations to exercise are compelling. Considering the discoveries in other areas of COX regulation of health and disease, there is certainly an interesting future of investigation in this re-emerging area, especially as it pertains to older individuals and the condition of sarcopenia, as well as exercise training and performance of individuals of all ages.
Keywords: PGE2; PGF2α; acetaminophen; ibuprofen; sarcopenia.
Figures

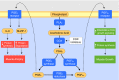
References
-
- Abramovitz M, Boie Y, Nguyen T, Rushmore TH, Bayne MA, Metters KM, Slipetz DM, Grygorczyk R. Cloning and expression of a cDNA for the human prostanoid FP receptor. J Biol Chem 269: 2632–2636, 1994 - PubMed
-
- Alkner BA, Tesch PA. Knee extensor and plantar flexor muscle size and function following 90 days of bed rest with or without resistance exercise. Eur J Appl Physiol 93: 294–305, 2004 - PubMed
-
- Aronoff DM. Aspirin and Reye’s syndrome: discovery of aspirin and paracetamol. Drug Saf 25: 751, 2002 - PubMed
-
- Baracos V, Rodemann HP, Dinarello CA, Goldberg AL. Stimulation of muscle protein degradation and prostaglandin E2 release by leukocytic pyrogen (interleukin-1). A mechanism for the increased degradation of muscle proteins during fever. N Engl J Med 308: 553–558, 1983 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

