Bioreactor design for perfusion-based, highly-vascularized organ regeneration
- PMID: 23542907
- PMCID: PMC3610919
- DOI: 10.1016/j.coche.2012.12.001
Bioreactor design for perfusion-based, highly-vascularized organ regeneration
Abstract
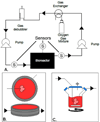
Bioartificial or laboratory-grown organs is a growing field centered on developing replacement organs and tissues to restore body function and providing a potential solution to the shortage of donor organs for transplantation. With the entry of engineered planar tissues, such as bladder and trachea, into clinical studies, an increasing focus is being given to designing complex, three-dimensional solid organs. As tissues become larger, thicker and more complex, the vascular network becomes crucial for supplying nutrients and maintaining viability and growth of the neo-organ. Perfusion decellularization, the process of removing cells from an entire organ, leaves the matrix of the vascular network intact. Organ engineering requires a delicate process of decellularization, sterilization, reseeding with appropriate cells, and organ maturation and stimulation to ensure optimal development. The design of bioreactors to facilitate this sequence of events has been refined to the extent that some bioartificial organs grown in these systems have been transplanted into recipient animals with sustained, though limited, function. This review focuses on the state-of-art in bioreactor development for perfusion-based bioartificial organs and highlights specific design components in need of further refinement.
Keywords: Bioreactors; Tissue engineering; bioartificial organs; organ transplantation; perfusion decellularization; regenerative medicine.
Figures

References
-
- Arcasoy SM, Kotloff RM. Lung transplantation. The New England journal of medicine. 1999;340(14):1081–1091. - PubMed
-
- Wertheim JA, Petrowsky H, Saab S, Kupiec-Weglinski JW, Busuttil RW. Major challenges limiting liver transplantation in the united states. American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons. 2011;11(9):1773–1784. - PMC - PubMed
-
- Organ procurement and transplantation network. [Accessed December 1, 2012]; http://optn.transplant.hrsa.gov/.
-
- Baptista PM, Siddiqui MM, Lozier G, Rodriguez SR, Atala A, Soker S. The use of whole organ decellularization for the generation of a vascularized liver organoid. Hepatology. 2011;53(2):604–617. - PubMed
-
-
Soto-gutierrez A, Zhang L, Medberry C, Fukumitsu K, Faulk D, Jiang H, Reing J, Gramignoli R, Komori J, Ross M, Nagaya M, et al. A whole-organ regenerative medicine approach for liver replacement. Tissue engineering Part C, Methods. 2011;17(6):677–689. This paper evaluates different methods to recellularize the liver parenchyma of rats. Direct injection of hepatocytes in divided doses is shown to be the optimal method.
-
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
