Electrochemical characterization of globotriose-containing self-assembled monolayers on nanoporous gold and their binding of soybean agglutinin
- PMID: 23545324
- PMCID: PMC3615452
- DOI: 10.1016/j.carres.2012.09.021
Electrochemical characterization of globotriose-containing self-assembled monolayers on nanoporous gold and their binding of soybean agglutinin
Abstract
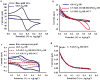
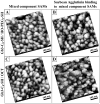
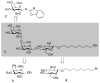
Self-assembled monolayers (SAMs) of α-D-Gal-(1→4)-β-D-Gal-(1→4)-β-D-Glc-mercaptooctane (globotriose, Gb3-C8-SH) were prepared both as single-component SAMs and as mixed SAMs with either octanethiol (OCT) or 8-mercapto-3,6-dioxaoctanol (HO-PEG2-SH), on flat gold and on nanoporous gold (NPG) electrodes. The binding of soybean agglutinin (SBA) to the globotriose (Gb3) unit in the SAMs was then studied using electrochemical impedance spectroscopy (EIS), which is a label free method found to be quite sensitive to SAM composition and to the differences in SAM structure on NPG versus on flat Au. The affinity of SBA to the mixed SAM of HO-PEG2-SH and Gb3-C8-SH on NPG is found to be greater on NPG than on flat gold, and indicates a potential advantage for NPG as a substrate. The SAMs of HO-PEG2-SH were found to resist protein adsorption on either NPG or flat gold. The non-specific adsorption of SBA to OCT SAMs on flat Au was observed in EIS by the increase in charge transfer resistance; whereas, the increase seen on the NPG surface was smaller, and suggests that EIS measurements on NPG are less affected by non-specific protein adsorption. Atomic force microscopy (AFM) images of the SBA binding to mixed SAM of HO-PEG2-SH and Gb3-C8-SH on NPG showed a greater number of proteins on top of the OCT containing SAMs.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures



Similar articles
-
Lectin-carbohydrate interactions on nanoporous gold monoliths.New J Chem. 2013 Jul 1;37(7):2150-2165. doi: 10.1039/C3NJ00253E. New J Chem. 2013. PMID: 24883017 Free PMC article.
-
Comparative Study of the Binding of Concanavalin A to Self-Assembled Monolayers Containing a Thiolated α-Mannoside on Flat Gold and on Nanoporous Gold.J Carbohydr Chem. 2012 Jan 1;31(4-6):466-503. doi: 10.1080/07328303.2012.683909. Epub 2012 Jul 2. J Carbohydr Chem. 2012. PMID: 23519474 Free PMC article.
-
Globotriose- and oligo(ethylene glycol)-terminated self-assembled monolayers: surface forces, wetting, and surfactant adsorption.Langmuir. 2006 Nov 21;22(24):10038-46. doi: 10.1021/la061857y. Langmuir. 2006. PMID: 17106997
-
Some thoughts on the existence of ion and water channels in highly dense and well-ordered CH3-terminated alkanethiol self-assembled monolayers on gold.Biosens Bioelectron. 2009 Jan 1;24(5):1074-82. doi: 10.1016/j.bios.2008.08.034. Epub 2008 Aug 28. Biosens Bioelectron. 2009. PMID: 18849157 Review.
-
Using self-assembled monolayers to understand the interactions of man-made surfaces with proteins and cells.Annu Rev Biophys Biomol Struct. 1996;25:55-78. doi: 10.1146/annurev.bb.25.060196.000415. Annu Rev Biophys Biomol Struct. 1996. PMID: 8800464 Review.
Cited by
-
Lectin-carbohydrate interactions on nanoporous gold monoliths.New J Chem. 2013 Jul 1;37(7):2150-2165. doi: 10.1039/C3NJ00253E. New J Chem. 2013. PMID: 24883017 Free PMC article.
-
Electrochemical Impedance Spectroscopy Study of Concanavalin A Binding to Self-Assembled Monolayers of Mannosides on Gold Wire Electrodes.J Electroanal Chem (Lausanne). 2016 Nov 1;780:311-320. doi: 10.1016/j.jelechem.2016.09.045. Epub 2016 Sep 28. J Electroanal Chem (Lausanne). 2016. PMID: 28413372 Free PMC article.
-
Are glycan biosensors an alternative to glycan microarrays?Anal Methods. 2014 Sep 7;6(17):6610-6620. doi: 10.1039/C4AY00692E. Epub 2014 Jul 9. Anal Methods. 2014. PMID: 27231487 Free PMC article.
References
-
- Zhi Z-l, Powell AK, Turnbull JE. Anal Chem. 2006;78:4786–4793. - PubMed
-
- Revell DJ, Knight JR, Blyth DJ, Haines AH, Russell DA. Langmuir. 1998;14:4517–4524.
-
- Smith EA, Thomas WD, Kiessling LL, Corn RM. J Am Chem Soc. 2003;125:6140–6148. - PubMed
-
- Mrksich M, Whitesides GM. Annu Rev Biophys Biomol Struct. 1996;25:55–78. - PubMed
-
- Zhang Y, Luo S, Tang Y, Yu L, Hou K-Y, Cheng J-P, Zeng X, Wang PG. Anal Chem. 2006;78:2001–2008. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

