Mechanism of origin DNA recognition and assembly of an initiator-helicase complex by SV40 large tumor antigen
- PMID: 23545501
- PMCID: PMC3748285
- DOI: 10.1016/j.celrep.2013.03.002
Mechanism of origin DNA recognition and assembly of an initiator-helicase complex by SV40 large tumor antigen
Abstract
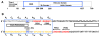
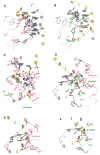
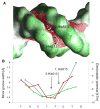
The DNA tumor virus Simian virus 40 (SV40) is a model system for studying eukaryotic replication. SV40 large tumor antigen (LTag) is the initiator/helicase that is essential for genome replication. LTag recognizes and assembles at the viral replication origin. We determined the structure of two multidomain LTag subunits bound to origin DNA. The structure reveals that the origin binding domains (OBDs) and Zn and AAA+ domains are involved in origin recognition and assembly. Notably, the OBDs recognize the origin in an unexpected manner. The histidine residues of the AAA+ domains insert into a narrow minor groove region with enhanced negative electrostatic potential. Computational analysis indicates that this region is intrinsically narrow, demonstrating the role of DNA shape readout in origin recognition. Our results provide important insights into the assembly of the LTag initiator/helicase at the replication origin and suggest that histidine contacts with the minor groove serve as a mechanism of DNA shape readout.
Copyright © 2013 The Authors. Published by Elsevier Inc. All rights reserved.
Figures
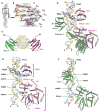
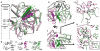
References
-
- Borowiec JA, Dean FB, Bullock PA, Hurwitz J. Binding and unwinding—how T antigen engages the SV40 origin of DNA replication. Cell. 1990;60:181–184. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

