Resetting translational homeostasis restores myelination in Charcot-Marie-Tooth disease type 1B mice
- PMID: 23547100
- PMCID: PMC3620355
- DOI: 10.1084/jem.20122005
Resetting translational homeostasis restores myelination in Charcot-Marie-Tooth disease type 1B mice
Abstract
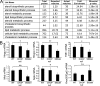
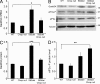
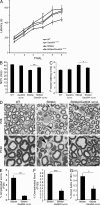
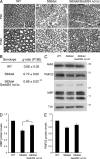
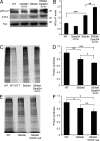
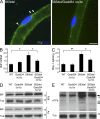
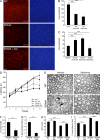
P0 glycoprotein is an abundant product of terminal differentiation in myelinating Schwann cells. The mutant P0S63del causes Charcot-Marie-Tooth 1B neuropathy in humans, and a very similar demyelinating neuropathy in transgenic mice. P0S63del is retained in the endoplasmic reticulum of Schwann cells, where it promotes unfolded protein stress and elicits an unfolded protein response (UPR) associated with translational attenuation. Ablation of Chop, a UPR mediator, from S63del mice completely rescues their motor deficit and reduces active demyelination by half. Here, we show that Gadd34 is a detrimental effector of CHOP that reactivates translation too aggressively in myelinating Schwann cells. Genetic or pharmacological limitation of Gadd34 function moderates translational reactivation, improves myelination in S63del nerves, and reduces accumulation of P0S63del in the ER. Resetting translational homeostasis may provide a therapeutic strategy in tissues impaired by misfolded proteins that are synthesized during terminal differentiation.
Figures

References
-
- Arthur-Farraj P.J., Latouche M., Wilton D.K., Quintes S., Chabrol E., Banerjee A., Woodhoo A., Jenkins B., Rahman M., Turmaine M., et al. 2012. c-Jun reprograms Schwann cells of injured nerves to generate a repair cell essential for regeneration. Neuron. 75:633–647 10.1016/j.neuron.2012.06.021 - DOI - PMC - PubMed
-
- Back S.H., Scheuner D., Han J., Song B., Ribick M., Wang J., Gildersleeve R.D., Pennathur S., Kaufman R.J. 2009. Translation attenuation through eIF2alpha phosphorylation prevents oxidative stress and maintains the differentiated state in beta cells. Cell Metab. 10:13–26 10.1016/j.cmet.2009.06.002 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

