Sex steroid hormones regulate constitutive expression of Cyp2e1 in female mouse liver
- PMID: 23548611
- PMCID: PMC3651618
- DOI: 10.1152/ajpendo.00585.2012
Sex steroid hormones regulate constitutive expression of Cyp2e1 in female mouse liver
Abstract
CYP2E1 is of paramount toxicological significance because it metabolically activates a large number of low-molecular-weight toxicants and carcinogens. In this context, factors that interfere with Cyp2e1 regulation may critically affect xenobiotic toxicity and carcinogenicity. The aim of this study was to investigate the role of female steroid hormones in the regulation of CYP2E1, as estrogens and progesterone are the bases of contraceptives and hormonal replacement therapy in menopausal women. Interestingly, a fluctuation in the hepatic expression pattern of Cyp2e1 was revealed in the different phases of the estrous cycle of female mice, with higher Cyp2e1 expression at estrus (E) and lower at methestrus (ME), highly correlated with that in plasma gonadal hormone levels. Depletion of sex steroids by ovariectomy repressed Cyp2e1 expression to levels similar to those detected in males and cyclic females at ME. Hormonal supplementation brought Cyp2e1 expression back to levels detected at E. The role of progesterone appeared to be more prominent than that of 17β-estradiol. Progesterone-induced Cyp2e1 upregulation could be attributed to inactivation of the insulin/PI3K/Akt/FOXO1 signaling pathway. Tamoxifen, an anti-estrogen, repressed Cyp2e1 expression potentially via activation of the PI3K/Akt/FOXO1 and GH/STAT5b-linked pathways. The sex steroid hormone-related changes in hepatic Cyp2e1 expression were highly correlated with those observed in Hnf-1α, β-catenin, and Srebp-1c. In conclusion, female steroid hormones are clearly involved in the regulation of CYP2E1, thus affecting the metabolism of a plethora of toxicants and carcinogenic agents, conditions that may trigger several pathologies or exacerbate the outcomes of various pathophysiological states.
Keywords: 17β-estradiol; Cyp2e1; estrous cycle; mice; progesterone.
Figures
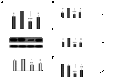


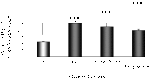






Similar articles
-
Sex steroid hormones differentially regulate CYP2D in female wild-type and CYP2D6-humanized mice.J Endocrinol. 2020 May;245(2):301-314. doi: 10.1530/JOE-19-0561. J Endocrinol. 2020. PMID: 32171179 Free PMC article.
-
D₂-dopaminergic receptor-linked pathways: critical regulators of CYP3A, CYP2C, and CYP2D.Mol Pharmacol. 2012 Oct;82(4):668-78. doi: 10.1124/mol.112.078709. Epub 2012 Jul 6. Mol Pharmacol. 2012. PMID: 22772593
-
Protective action of liraglutide in beta cells under lipotoxic stress via PI3K/Akt/FoxO1 pathway.J Cell Biochem. 2014 Jun;115(6):1166-75. doi: 10.1002/jcb.24763. J Cell Biochem. 2014. PMID: 24415347
-
Sex steroid hormones, the estrous cycle, and rapid modulation of glutamatergic synapse properties in the striatal brain regions with a focus on 17β-estradiol and the nucleus accumbens.Steroids. 2024 Jan;201:109344. doi: 10.1016/j.steroids.2023.109344. Epub 2023 Nov 17. Steroids. 2024. PMID: 37979822 Free PMC article. Review.
-
The interplay of sex steroid hormones and microRNAs in endometrial cancer: current understanding and future directions.Front Endocrinol (Lausanne). 2023 Apr 21;14:1166948. doi: 10.3389/fendo.2023.1166948. eCollection 2023. Front Endocrinol (Lausanne). 2023. PMID: 37152960 Free PMC article. Review.
Cited by
-
Stress as a Potential Regulatory Factor in the Outcome of Pharmacotherapy.Front Neurosci. 2022 Mar 23;16:737716. doi: 10.3389/fnins.2022.737716. eCollection 2022. Front Neurosci. 2022. PMID: 35401076 Free PMC article. No abstract available.
-
Ethanol potentiates the genotoxicity of the food-derived mammary carcinogen PhIP in human estrogen receptor-positive mammary cells: mechanistic support for lifestyle factors (cooked red meat and ethanol) associated with mammary cancer.Arch Toxicol. 2018 Apr;92(4):1639-1655. doi: 10.1007/s00204-018-2160-9. Epub 2018 Jan 23. Arch Toxicol. 2018. PMID: 29362861 Free PMC article.
-
Development of an IgY Antibody-Based Immunoassay for the Screening of the CYP2E1 Inhibitor/Enhancer from Herbal Medicines.Front Pharmacol. 2016 Dec 22;7:502. doi: 10.3389/fphar.2016.00502. eCollection 2016. Front Pharmacol. 2016. PMID: 28066249 Free PMC article.
-
Age-related modifications in CYP-dependent drug metabolism: role of stress.Front Endocrinol (Lausanne). 2023 May 24;14:1143835. doi: 10.3389/fendo.2023.1143835. eCollection 2023. Front Endocrinol (Lausanne). 2023. PMID: 37293497 Free PMC article. Review.
-
Cytochrome P450-2E1 is involved in aging-related kidney damage in mice through increased nitroxidative stress.Food Chem Toxicol. 2017 Nov;109(Pt 1):48-59. doi: 10.1016/j.fct.2017.08.022. Epub 2017 Aug 24. Food Chem Toxicol. 2017. PMID: 28843596 Free PMC article.
References
-
- Assaf S, Hazard D, Pitel F, Morisson M, Alizadeh M, Gondret F, Diot C, Vignal A, Douaire M, Lagarrigue S. Cloning of cDNA encoding the nuclear form of chicken sterol response element binding protein-2 (SREBP-2), chromosomal localization, and tissue expression of chicken SREBP-1 and -2 genes. Poult Sci 82: 54–61, 2003 - PubMed
-
- Akiyama TE, Gonzalez FJ. Regulation of P450 genes by liver-enriched transcription factors and nuclear receptors. Biochim Biophys Acta 1619: 223–234, 2003 - PubMed
-
- Banerjee S, Shang TQ, Wilson AM, Moore AL, Strand SE, Gordon MP, Doty SL. Expression of functional mammalian P450 2E1 in hairy root cultures. Biotechnol Bioeng 77: 462–466, 2002 - PubMed
-
- Baynes JW, Dominiczak MH. Medical Biochemistry (2nd ed). Philadelphia, PA: Elsevier Mosby, p. 555, 2005
-
- Bebia Z, Buch SC, Wilson JW, Frye RF, Romkes M, Cecchetti A, Chaves-Gnecco D, Branch RA. Bioequivalence revisited: influence of age and sex on CYP enzymes. Clin Pharmacol Ther 76: 618–627, 2004 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

