Vitrification preserves chromatin integrity, bioenergy potential and oxidative parameters in mouse embryos
- PMID: 23552480
- PMCID: PMC3652727
- DOI: 10.1186/1477-7827-11-27
Vitrification preserves chromatin integrity, bioenergy potential and oxidative parameters in mouse embryos
Abstract
Background: The aim of this study was to evaluate the effects of vitrification on morpho-functional parameters (blastomere/chromatin integrity and bioenergy/oxidative potential) of mouse preimplantation embryos.
Methods: In vivo produced mouse (4/16-cell, morulae and blastocyst-stage) embryos were randomly divided into vitrification and control groups. For vitrification, embryos were exposed to a 2-step loading of ethylene glycol and propylene glycol, before being placed in a small nylon loop and submerged into liquid nitrogen. After warming, the cryoprotectants were diluted by a 3-step procedure. Embryo morphology, chromatin integrity and energy/oxidative status were compared between groups.
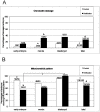
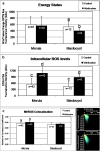
Results: Vitrification induced low grade blastomere cytofragmentation (P < 0.05) and low chromatin damage only in embryos at the morula stage (P < 0.001). Mitochondrial (mt) distribution pattern was affected by vitrification only in early embryos (P < 0.001). Mitochondrial activity did not change upon vitrification in morula-stage embryos but it was reduced in blastocyst-stage embryos (P < 0.05). Intracellular ROS levels significantly increased in embryos at the morula and blastocyst stages (P < 0.001). Colocalization of active mitochondria and ROS increased only in vitrified blastocysts.
Conclusions: In conclusion, this study elucidates the developmentally-related and mild effects of vitrification on morphology, nuclear and bioenergy/oxidative parameters of mouse embryos and demonstrates that vitrification is a suitable method for preserving predictive parameters of embryo ability to induce a full-term pregnancy.
Figures

References
-
- Fuller B, Paynter S, Watson P. In: Life in the frozen state. Fuller B, Lane N, Benson E, editor. Boca Raton: CRC Press; 2004. Cryopreservation of human gametes and embryos; pp. 505–541.
-
- Vajta G. Vitrification of oocytes and embryos of domestic animals. Anim Reprod Sci. 2000;60–61:357–364. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

