Rapid quantification of pixel-wise fiber orientation data in micrographs
- PMID: 23552635
- PMCID: PMC3639785
- DOI: 10.1117/1.JBO.18.4.046003
Rapid quantification of pixel-wise fiber orientation data in micrographs
Abstract
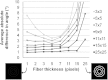
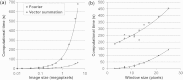
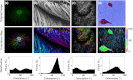
Defining fiber orientation at each pixel within a medical image has traditionally been computationally intensive and prone to systematic errors. A weighted orientation vector summation algorithm capable of detecting fiber orientation simultaneously at each pixel within an image is presented. As a result, pixel-specific fiber orientation information with 2 deg to 3 deg accuracy can be determined within seconds, enabling the practical use of pixel-wise orientation data for characterizing structural anisotropy. This analysis technique has applicability and potential diagnostic utility for a variety of modalities, including second harmonic generation, scanning electron microscopy and immunohistochemical imaging is demonstrated.
Figures


Similar articles
-
Rapid three-dimensional quantification of voxel-wise collagen fiber orientation.Biomed Opt Express. 2015 Jun 3;6(7):2294-310. doi: 10.1364/BOE.6.002294. eCollection 2015 Jul 1. Biomed Opt Express. 2015. PMID: 26203362 Free PMC article.
-
Comparison of 2D fiber network orientation measurement methods.J Biomed Mater Res A. 2009 Feb;88(2):322-31. doi: 10.1002/jbm.a.31847. J Biomed Mater Res A. 2009. PMID: 18286605
-
Methods for Quantifying Fibrillar Collagen Alignment.Methods Mol Biol. 2017;1627:429-451. doi: 10.1007/978-1-4939-7113-8_28. Methods Mol Biol. 2017. PMID: 28836218 Free PMC article.
-
Fundamentals of image processing in light microscopy.Methods Cell Biol. 2003;72:217-42. doi: 10.1016/s0091-679x(03)72011-1. Methods Cell Biol. 2003. PMID: 14719334 Review. No abstract available.
-
Quantitative fluorescence microscopy and image deconvolution.Methods Cell Biol. 2003;72:349-67. doi: 10.1016/s0091-679x(03)72017-2. Methods Cell Biol. 2003. PMID: 14719340 Review. No abstract available.
Cited by
-
Highly accurate, automated quantification of 2D/3D orientation for cerebrovasculature using window optimizing method.J Biomed Opt. 2022 Oct;27(10):105003. doi: 10.1117/1.JBO.27.10.105003. J Biomed Opt. 2022. PMID: 36273250 Free PMC article.
-
The zonal evolution of collagen-network morphology quantified in early osteoarthritic grades of human cartilage.Osteoarthr Cartil Open. 2020 Jul 28;2(4):100086. doi: 10.1016/j.ocarto.2020.100086. eCollection 2020 Dec. Osteoarthr Cartil Open. 2020. PMID: 36474870 Free PMC article.
-
Characterizing differences in the collagen fiber organization of skin wounds using quantitative polarized light imaging.Wound Repair Regen. 2019 Nov;27(6):711-714. doi: 10.1111/wrr.12758. Epub 2019 Sep 3. Wound Repair Regen. 2019. PMID: 31418977 Free PMC article.
-
Three-dimensional collagen fiber mapping and tractography of human uterine tissue using OCT.Biomed Opt Express. 2020 Sep 11;11(10):5518-5541. doi: 10.1364/BOE.397041. eCollection 2020 Oct 1. Biomed Opt Express. 2020. PMID: 33149968 Free PMC article.
-
Conditional deletion of RB1 in the Tie2 lineage leads to aortic valve regurgitation.PLoS One. 2018 Jan 5;13(1):e0190623. doi: 10.1371/journal.pone.0190623. eCollection 2018. PLoS One. 2018. PMID: 29304157 Free PMC article.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

