Skeletal muscle hypertrophy and regeneration: interplay between the myogenic regulatory factors (MRFs) and insulin-like growth factors (IGFs) pathways
- PMID: 23552962
- PMCID: PMC11113627
- DOI: 10.1007/s00018-013-1330-4
Skeletal muscle hypertrophy and regeneration: interplay between the myogenic regulatory factors (MRFs) and insulin-like growth factors (IGFs) pathways
Abstract
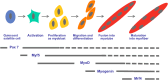
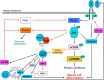
Adult skeletal muscle can regenerate in response to muscle damage. This ability is conferred by the presence of myogenic stem cells called satellite cells. In response to stimuli such as injury or exercise, these cells become activated and express myogenic regulatory factors (MRFs), i.e., transcription factors of the myogenic lineage including Myf5, MyoD, myogenin, and Mrf4 to proliferate and differentiate into myofibers. The MRF family of proteins controls the transcription of important muscle-specific proteins such as myosin heavy chain and muscle creatine kinase. Different growth factors are secreted during muscle repair among which insulin-like growth factors (IGFs) are the only ones that promote both muscle cell proliferation and differentiation and that play a key role in muscle regeneration and hypertrophy. Different isoforms of IGFs are expressed during muscle repair: IGF-IEa, IGF-IEb, or IGF-IEc (also known as mechano growth factor, MGF) and IGF-II. MGF is expressed first and is observed in satellite cells and in proliferating myoblasts whereas IGF-Ia and IGF-II expression occurs at the state of muscle fiber formation. Interestingly, several studies report the induction of MRFs in response to IGFs stimulation. Inversely, IGFs expression may also be regulated by MRFs. Various mechanisms are proposed to support these interactions. In this review, we describe the general process of muscle hypertrophy and regeneration and decipher the interactions between the two groups of factors involved in the process.
Figures

References
-
- Schmalbruch H, Lewis DM. Dynamics of nuclei of muscle fibers and connective tissue cells in normal and denervated rat muscles. Muscle Nerve. 2000;23:617–626. - PubMed
-
- Irintchev A, Wernig A. Muscle damage and repair in voluntarily running mice: strain and muscle differences. Cell Tissue Res. 1987;249:509–521. - PubMed
-
- Papadimitriou JM, Robertson TA, Mitchell CA, Grounds MD. The process of new plasmalemma formation in focally injured skeletal muscle fibers. J Struct Biol. 1990;103:124–134. - PubMed
-
- Charge SB, Rudnicki MA. Cellular and molecular regulation of muscle regeneration. Physiol Rev. 2004;84:209–238. - PubMed
-
- Carlson BM, Faulkner JA. The regeneration of skeletal muscle fibers following injury: a review. Med Sci Sports Exerc. 1983;15:187–198. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

