Ethylene Response Factor6 acts as a central regulator of leaf growth under water-limiting conditions in Arabidopsis
- PMID: 23553636
- PMCID: PMC3641212
- DOI: 10.1104/pp.113.216341
Ethylene Response Factor6 acts as a central regulator of leaf growth under water-limiting conditions in Arabidopsis
Abstract
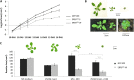
Leaf growth is a complex developmental process that is continuously fine-tuned by the environment. Various abiotic stresses, including mild drought stress, have been shown to inhibit leaf growth in Arabidopsis (Arabidopsis thaliana), but the underlying mechanisms remain largely unknown. Here, we identify the redundant Arabidopsis transcription factors ETHYLENE RESPONSE FACTOR5 (ERF5) and ERF6 as master regulators that adapt leaf growth to environmental changes. ERF5 and ERF6 gene expression is induced very rapidly and specifically in actively growing leaves after sudden exposure to osmotic stress that mimics mild drought. Subsequently, enhanced ERF6 expression inhibits cell proliferation and leaf growth by a process involving gibberellin and DELLA signaling. Using an ERF6-inducible overexpression line, we demonstrate that the gibberellin-degrading enzyme GIBBERELLIN 2-OXIDASE6 is transcriptionally induced by ERF6 and that, consequently, DELLA proteins are stabilized. As a result, ERF6 gain-of-function lines are dwarfed and hypersensitive to osmotic stress, while the growth of erf5erf6 loss-of-function mutants is less affected by stress. Besides its role in plant growth under stress, ERF6 also activates the expression of a plethora of osmotic stress-responsive genes, including the well-known stress tolerance genes STZ, MYB51, and WRKY33. Interestingly, activation of the stress tolerance genes by ERF6 occurs independently from the ERF6-mediated growth inhibition. Together, these data fit into a leaf growth regulatory model in which ERF5 and ERF6 form a missing link between the previously observed stress-induced 1-aminocyclopropane-1-carboxylic acid accumulation and DELLA-mediated cell cycle exit and execute a dual role by regulating both stress tolerance and growth inhibition.
Figures





References
-
- Achard P, Gusti A, Cheminant S, Alioua M, Dhondt S, Coppens F, Beemster GTS, Genschik P. (2009) Gibberellin signaling controls cell proliferation rate in Arabidopsis. Curr Biol 19: 1188–1193 - PubMed
-
- Alonso JM, Stepanova AN, Leisse TJ, Kim CJ, Chen H, Shinn P, Stevenson DK, Zimmerman J, Barajas P, Cheuk R, et al. (2003) Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 301: 653–657 - PubMed
-
- Anastasiou E, Lenhard M. (2007) Growing up to one’s standard. Curr Opin Plant Biol 10: 63–69 - PubMed
-
- Andriankaja M, Dhondt S, De Bodt S, Vanhaeren H, Coppens F, De Milde L, Mühlenbock P, Skirycz A, Gonzalez N, Beemster GTS, et al. (2012) Exit from proliferation during leaf development in Arabidopsis thaliana: a not-so-gradual process. Dev Cell 22: 64–78 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

