Distinct prestimulus and poststimulus activation of VTA neurons correlates with stimulus detection
- PMID: 23554430
- PMCID: PMC3727034
- DOI: 10.1152/jn.00784.2012
Distinct prestimulus and poststimulus activation of VTA neurons correlates with stimulus detection
Abstract
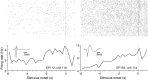
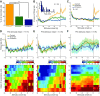
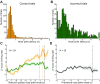
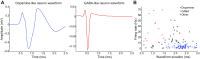
Dopamine neurons of the ventral tegmental area (VTA) signal the occurrence of a reward-predicting conditioned stimulus (CS) with a subsecond duration increase in post-CS firing rate. Important theories about reward-prediction error and reward expectancy have been informed by the substantial number of studies that have examined post-CS phasic VTA neuron activity. On the other hand, the role of VTA neurons in anticipation of a reward-predicting CS and analysis of prestimulus spike rate rarely has been studied. We recorded from the VTA in rats during the 3-choice reaction time task, which has a fixed-duration prestimulus period and a difficult-to-detect stimulus. Use of a stimulus that was difficult to detect led to behavioral errors, which allowed us to compare VTA activity between trials with correct and incorrect stimulus-guided choices. We found a sustained increase in firing rate of both putative dopamine and GABA neurons during the pre-CS period of correct and incorrect trials. The poststimulus phasic response, however, was absent on incorrect trials, suggesting that the stimulus-evoked phasic response of dopamine neurons may relate to stimulus detection. The prestimulus activation of VTA neurons may modulate cortical systems that represent internal states of stimulus expectation and provide a mechanism for dopamine neurotransmission to influence preparatory attention to an expected stimulus.
Keywords: ADHD; attention; dopamine; prefrontal cortex; reward; schizophrenia.
Figures


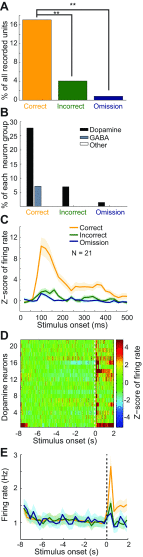
References
-
- Apicella P, Scarnati E, Ljungberg T, Schultz W. Neuronal activity in monkey striatum related to the expectation of predictable environmental events. J Neurophysiol 68: 945–960, 1992 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

