Ex vivo modeling of chemical synergy in prenatal kidney cystogenesis
- PMID: 23554868
- PMCID: PMC3595278
- DOI: 10.1371/journal.pone.0057797
Ex vivo modeling of chemical synergy in prenatal kidney cystogenesis
Abstract
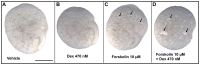
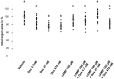
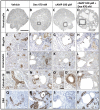
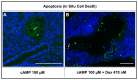
Cyclic adenosine monophosphate (cAMP) drives genetic polycystic kidney disease (PKD) cystogenesis. Yet within certain PKD families, striking differences in disease severity exist between affected individuals, and genomic and/or environmental modifying factors have been evoked to explain these observations. We hypothesized that PKD cystogenesis is accentuated by an aberrant fetal milieu, specifically by glucocorticoids. The extent and nature of cystogenesis was assessed in explanted wild-type mouse embryonic metanephroi, using 8-Br-cAMP as a chemical to mimic genetic PKD and the glucocorticoid dexamethasone as the environmental modulator. Cysts and glomeruli were quantified by an observer blinded to culture conditions, and tubules were phenotyped using specific markers. Dexamethasone or 8-Br-cAMP applied on their own produced cysts predominantly arising in proximal tubules and descending limbs of loops of Henle. When applied together, however, dexamethasone over a wide concentration range synergized with 8-Br-cAMP to generate a more severe, glomerulocystic, phenotype; we note that prominent glomerular cysts have been reported in autosomal dominant PKD fetal kidneys. Our data support the idea that an adverse antenatal environment exacerbates renal cystogenesis.
Conflict of interest statement
Figures








References
-
- UK Renal Registry website. Available: http://www.renalreg.com/Reports/2006.html. Accessed 2013 Feb 13
-
- Harris PC (2009) 2008 Homer W. Smith Award: insights into the pathogenesis of polycystic kidney disease from gene discovery. J Am Soc Nephrol 20: 1188–1198. - PubMed
-
- Ward CJ, Hogan MC, Rossetti S, Walker D, Sneddon T, et al. (2002) The gene mutated in autosomal recessive polycystic kidney disease encodes a large, receptor-like protein. Nat Genet 2002; 30: 259–269. - PubMed
-
- Furu L, Onuchic LF, Gharavi A, Hou X, Esquivel EL, et al. (2003) Milder presentation of recessive polycystic kidney disease requires presence of amino acid substitution mutations. J Am Soc Nephrol 14: 2004–2014. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

