Silver(I) complex formation with cysteine, penicillamine, and glutathione
- PMID: 23556419
- PMCID: PMC3684387
- DOI: 10.1021/ic400192c
Silver(I) complex formation with cysteine, penicillamine, and glutathione
Abstract
The complex formation between silver(I) and cysteine (H2Cys), penicillamine (H2Pen), and glutathione (H3Glu) in alkaline aqueous solution was examined using extended X-ray absorption fine structure (EXAFS) and (109)Ag NMR spectroscopic techniques. The complexes formed in 0.1 mol dm(-3) Ag(I) solutions with cysteine and penicillamine were investigated for ligand/Ag(I) (L/Ag) mole ratios increasing from 2.0 to 10.0. For the series of cysteine solutions (pH 10-11) a mean Ag-S bond distance of 2.45 ± 0.02 Å consistently emerged, while for penicillamine (pH 9) the average Ag-S bond distance gradually increased from 2.40 to 2.44 ± 0.02 Å. EXAFS and (109)Ag NMR spectra of a concentrated Ag(I)-cysteine solution (C(Ag(I)) = 0.8 mol dm(-3), L/Ag = 2.2) showed a mean Ag-S bond distance of 2.47 ± 0.02 Å and δ((109)Ag) 1103 ppm, consistent with prevailing, partially oligomeric AgS3 coordinated species, while for penicillamine (C(Ag(I)) = 0.5 mol dm(-3), L/Ag = 2.0) the mean Ag-S bond distance of 2.40 ± 0.02 Å and δ((109)Ag) 922 ppm indicate that mononuclear AgS2 coordinated complexes dominate. For Ag(I)-glutathione solutions (C(Ag(I)) = 0.01 mol dm(-3), pH ∼11), mononuclear AgS2 coordinated species with a mean Ag-S bond distance of 2.36 ± 0.02 Å dominate for L/Ag mole ratios from 2.0 to 10.0. The crystal structure of the silver(I)-cysteine compound (NH4)Ag2(HCys)(Cys)·H2O (1) precipitating at pH ∼10 was solved and showed a layer structure with both AgS3 and AgS3N coordination to the cysteinate ligands. A redetermination of the crystal structure of Ag(HPen)·H2O (2) confirmed the proposed digonal AgS2 coordination environment to bridging thiolate sulfur atoms in polymeric intertwining chains forming a double helix. A survey of Ag-S bond distances for crystalline Ag(I) complexes with S-donor ligands in different AgS2, AgS2(O/N), and AgS3 coordination environments was used, together with a survey of (109)Ag NMR chemical shifts, to assist assignments of the Ag(I) coordination in solution.
Figures

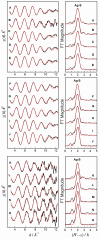
 ; Fit
; Fit  ) are presented in Table 5 (labelled with *).
) are presented in Table 5 (labelled with *).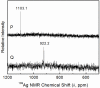
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

