Human neutrophil elastase-mediated goblet cell metaplasia is attenuated in TACE-deficient mice
- PMID: 23564510
- PMCID: PMC3652059
- DOI: 10.1152/ajplung.00259.2012
Human neutrophil elastase-mediated goblet cell metaplasia is attenuated in TACE-deficient mice
Abstract
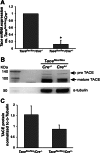
Neutrophilic inflammation is associated with chronic airway diseases. It has been observed that human neutrophil elastase (HNE), which is secreted by active neutrophils during inflammation, induces both mucin overproduction and goblet cell metaplasia. Several in vitro studies suggest that tumor necrosis factor-α converting enzyme (TACE) regulates the signaling axis that mediates HNE-induced mucin overproduction; however, it is unknown whether TACE performs a similar function in HNE-induced goblet cell metaplasia in vivo. We conducted this study to determine whether the inactivation of Tace gene expression attenuates HNE-induced goblet cell metaplasia in mice. Deletion of Tace is lethal shortly after birth in mice; therefore, we utilized Tace(flox/flox)R26CreER(+/-) mice and induced conditional deletion of Tace using a tamoxifen injection. Wild-type mice were given tamoxifen to control for its effect. Tace conditional deletion mice and wild-type mice were exposed to HNE via nasal instillation three times at 3-day intervals, and the lungs were harvested on day 11 after initial HNE exposure. Using periodic acid-Schiff staining and MUC5AC immunohistochemical staining to visualize goblet cells in the lungs, we found that HNE induced goblet cell metaplasia in the wild-type mice and that HNE-induced goblet cell metaplasia was significantly attenuated in the Tace conditional deletion mice. These findings suggest that TACE could be a potential target in the treatment of goblet cell metaplasia in patients with chronic airway diseases.
Keywords: airway epithelium; airway mucin; chronic airway disease; neutrophilic inflammation.
Figures



Similar articles
-
Human Neutrophil Elastase Mediates MUC5AC Hypersecretion via the Tumour Necrosis Factor-α Converting Enzyme-Epidermal Growth Factor Receptor Signalling Pathway in vivo.ORL J Otorhinolaryngol Relat Spec. 2021;83(5):310-318. doi: 10.1159/000509982. Epub 2021 Jun 15. ORL J Otorhinolaryngol Relat Spec. 2021. PMID: 34130299
-
Neutrophil elastase induces MUC5AC mucin production in human airway epithelial cells via a cascade involving protein kinase C, reactive oxygen species, and TNF-alpha-converting enzyme.J Immunol. 2005 Sep 15;175(6):4009-16. doi: 10.4049/jimmunol.175.6.4009. J Immunol. 2005. PMID: 16148149
-
Human neutrophil elastase induces MUC5AC overexpression in chronic rhinosinusitis through tumour necrosis factor-α converting enzyme.Acta Otolaryngol. 2016 Jun;136(6):641-8. doi: 10.3109/00016489.2016.1144145. Epub 2016 Feb 16. Acta Otolaryngol. 2016. PMID: 26881964
-
Role of neutrophils in mucus hypersecretion in COPD and implications for therapy.Treat Respir Med. 2004;3(3):147-59. doi: 10.2165/00151829-200403030-00003. Treat Respir Med. 2004. PMID: 15219174 Review.
-
Role of neutrophil elastase in hypersecretion during COPD exacerbations, and proposed therapies.Chest. 2000 May;117(5 Suppl 2):386S-9S. doi: 10.1378/chest.117.5_suppl_2.386s. Chest. 2000. PMID: 10843982 Review.
Cited by
-
Endoplasmic reticulum stress/XBP1 promotes airway mucin secretion under the influence of neutrophil elastase.Int J Mol Med. 2021 May;47(5):81. doi: 10.3892/ijmm.2021.4914. Epub 2021 Mar 24. Int J Mol Med. 2021. PMID: 33760106 Free PMC article.
-
Vinpocetine inhibits Streptococcus pneumoniae-induced upregulation of mucin MUC5AC expression via induction of MKP-1 phosphatase in the pathogenesis of otitis media.J Immunol. 2015 Jun 15;194(12):5990-8. doi: 10.4049/jimmunol.1401489. Epub 2015 May 13. J Immunol. 2015. PMID: 25972475 Free PMC article.
-
Expression and Clinical Significance of Mucin Gene in Chronic Rhinosinusitis.Curr Allergy Asthma Rep. 2020 Aug 18;20(11):63. doi: 10.1007/s11882-020-00958-w. Curr Allergy Asthma Rep. 2020. PMID: 32812123 Free PMC article. Review.
-
Putting the Squeeze on Airway Epithelia.Physiology (Bethesda). 2015 Jul;30(4):293-303. doi: 10.1152/physiol.00004.2015. Physiology (Bethesda). 2015. PMID: 26136543 Free PMC article. Review.
-
Glycosaminoglycans as Multifunctional Anti-Elastase and Anti-Inflammatory Drugs in Cystic Fibrosis Lung Disease.Front Pharmacol. 2020 Jul 8;11:1011. doi: 10.3389/fphar.2020.01011. eCollection 2020. Front Pharmacol. 2020. PMID: 32733248 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

