Hunger in the absence of caloric restriction improves cognition and attenuates Alzheimer's disease pathology in a mouse model
- PMID: 23565247
- PMCID: PMC3614512
- DOI: 10.1371/journal.pone.0060437
Hunger in the absence of caloric restriction improves cognition and attenuates Alzheimer's disease pathology in a mouse model
Abstract
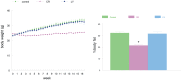
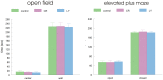
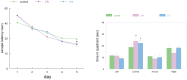
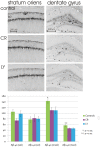
It has been shown that caloric restriction (CR) delays aging and possibly delays the development of Alzheimer's disease (AD). We conjecture that the mechanism may involve interoceptive cues, rather than reduced energy intake per se. We determined that hunger alone, induced by a ghrelin agonist, reduces AD pathology and improves cognition in the APP-SwDI mouse model of AD. Long-term treatment with a ghrelin agonist was sufficient to improve the performance in the water maze. The treatment also reduced levels of amyloid beta (Aβ) and inflammation (microglial activation) at 6 months of age compared to the control group, similar to the effect of CR. Thus, a hunger-inducing drug attenuates AD pathology, in the absence of CR, and the neuroendocrine aspects of hunger also prevent age-related cognitive decline.
Conflict of interest statement
Figures


References
-
- Wang J, Ho L, Qin W, Rocher AB, Seror I, et al. (2005) Caloric restriction attenuates beta-amyloid neuropathology in a mouse model of Alzheimer's disease. The FASEB journal: official publication of the Federation of American Societies for Experimental Biology 19: 659–661. - PubMed
-
- Wang J, Fivecoat H, Ho L, Pan Y, Ling E, et al. (2010) The role of Sirt1: at the crossroad between promotion of longevity and protection against Alzheimer's disease neuropathology. Biochim Biophys Acta 1804: ,1690-1694 . - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

