Modifications of longitudinally extensive transverse myelitis and brainstem lesions in the course of neuromyelitis optica (NMO): a population-based, descriptive study
- PMID: 23566260
- PMCID: PMC3622587
- DOI: 10.1186/1471-2377-13-33
Modifications of longitudinally extensive transverse myelitis and brainstem lesions in the course of neuromyelitis optica (NMO): a population-based, descriptive study
Abstract
Background: Neuromyelitis optica (NMO) includes transverse myelitis, optic neuritis and brain lesions. Recent studies have indicated that the brainstem is an important site of attack in NMO. Longitudinally extensive transverse myelitis (LETM) is an important component of the clinical diagnosis of NMO. The frequency of brainstem and LETM lesions, changes over time of LETM and the clinical consequences in the course of NMO have only been sparsely studied.
Methods: The study was a population-based retrospective case series with clinical and magnetic resonance imaging (MRI) follow-up of 35 patients with definite NMO and a relapsing-remitting course.
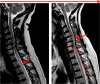
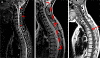
Results: Brainstem lesions were observed in 25 patients, 18 in medulla oblongata (11 in area postrema). Lesions in the pons, mesencephalon and diencephalon occurred in 10, 7 and 7 patients, respectively. Lesions were symptomatic in medulla oblongata and pons, asymptomatic in mesencephalon and diencephalon. Brainstem lesions were observed significantly more often in anti-aquaporin-4 (AQP-4) antibody positive than in seronegative patients (p < 0.002).LETM was demonstrated by MRI of the spinal cord in 30/36 patients, 23/30 of whom had follow-up MRI of the spinal cord. Recurrent LETM was observed in five patients. In nine patients the LETM changed into multiple lesions during remission or treatment. Spinal cord atrophy was observed in 12/23 (52%) patients, correlating to Expanded Disability Status Scale (r = 0.88, p < 0.001).
Conclusions: NMO patients had frequent occurrence of brainstem lesions and LETM. Brainstem lesions were associated with anti-AQP4 antibody positivity. LETM lesions differentiated over time and the outcome included relapses, fragmentation and atrophy. Correlation was observed between spinal cord atrophy and neurological disability.
Figures


References
-
- Cloys D, Netsky M. In: Handbook of clinical neurology. 9. Viken PJ BG, editor. North-Holland: Amsterdam; 1970. Neuromyelitis optica; pp. 426–436.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

