Safety and immunogenicity of 2010–2011 A/H1N1pdm09-containing trivalent inactivated influenza vaccine in adults previously given AS03-adjuvanted H1N1 2009 pandemic vaccine: results of a randomized trial
- PMID: 23570051
- PMCID: PMC3667929
- DOI: 10.4161/hv.22619
Safety and immunogenicity of 2010–2011 A/H1N1pdm09-containing trivalent inactivated influenza vaccine in adults previously given AS03-adjuvanted H1N1 2009 pandemic vaccine: results of a randomized trial
Abstract
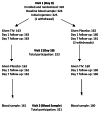
Many Canadians received a novel AS03-adjuvanted vaccine during the 2009 influenza A/H1N1 pandemic. Longer term implications of adjuvant use were unclear: would anti-H1N1 immune responses persist at high levels and, if so, could that result in increased or unusual adverse effects upon re-exposure to H1N1pdm09 antigen in the trivalent influenza vaccine (TIV) for 2010-11? To answer these questions, adults given AS03-adjuvanted H1N1pdm09 vaccine (Arepanrix®, GSK Canada) 9-10 mo earlier were enrolled in an evaluator-blinded, crossover trial to receive 2010-2011 non-adjuvanted TIV (Fluviral®, GSK Canada) and placebo 10 d apart, in random order. Adverse effects were monitored for 7 d after each injection. Vaccine-attributable adverse event (VAAE) rates were calculated by subtracting rates after placebo from those after vaccine. Blood was obtained pre-vaccination and 21-30 d afterward to measure hemagglutination inhibiting antibody titers. In total, 326 participants were enrolled and 321 completed the study. VAAE rates were low except for myalgia (18.6%) and injection site pain (63.2%). At baseline, H1N1pdm09 titers ≥ 40 were present in 176/325 subjects (54.2%, 95% confidence interval 48.6, 59.7), with a geometric mean titer (GMT) of 37.4 (95% CI 32.8, 42.6). Post-immunization, 96.0% (95% CI 92.3, 97.8) had H1N1pdm09 titers ≥ 40, with GMT of 167.4 (95% CI 148.7, 188.5). Responses to both influenza A strains in TIV were similar, implying no lasting effect of adjuvant exposure. In summary, titers ≥ 40 persisted in only half the participants 9-10 mo after adjuvanted pandemic vaccine but were restored in nearly all after TIV vaccination, with minimal increase in adverse effects.
Figures
References
-
- Roman F, Vaman T, Gerlach B, Markendorf A, Gillard P, Devaster JM. Immunogenicity and safety in adults of one dose of influenza A H1N1v 2009 vaccine formulated with and without AS03A-adjuvant: preliminary report of an observer-blind, randomised trial. Vaccine. 2010;28:1740–5. doi: 10.1016/j.vaccine.2009.12.014. - DOI - PubMed
-
- Scheifele DW, Ward BJ, Dionne M, Vanderkooi O, Loeb M, Coleman BL, et al. Compatibility of AS03-adjuvanted A/H1N1pdm09 and seasonal trivalent influenza vaccines in adults: results of a randomized, controlled trial. Vaccine. In press. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical