Towards polypharmacokinetics: pharmacokinetics of multicomponent drugs and herbal medicines using a metabolomics approach
- PMID: 23573155
- PMCID: PMC3612473
- DOI: 10.1155/2013/819147
Towards polypharmacokinetics: pharmacokinetics of multicomponent drugs and herbal medicines using a metabolomics approach
Abstract
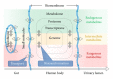
Determination of pharmacokinetics (PKs) of multicomponent pharmaceuticals and/or nutraceuticals (polypharmacokinetics, poly-PKs) is difficult due to the vast number of compounds present in natural products, their various concentrations across a wide range, complexity of their interactions, as well as their complex degradation dynamics in vivo. Metabolomics coupled with multivariate statistical tools that focus on the comprehensive analysis of small molecules in biofluids is a viable approach to address the challenges of poly-PK. This paper discusses recent advances in the characterization of poly-PK and the metabolism of multicomponent xenobiotic agents, such as compound drugs, dietary supplements, and herbal medicines, using metabolomics strategy. We propose a research framework that integrates the dynamic concentration profile of bioavailable xenobiotic molecules that result from in vivo absorption and hepatic and gut bacterial metabolism, as well as the human metabolic response profile. This framework will address the bottleneck problem in the pharmacological evaluation of multicomponent pharmaceuticals and nutraceuticals, leading to the direct elucidation of the pharmacological and molecular mechanisms of these compounds.
Figures

Similar articles
-
Poly-pharmacokinetic Study of a Multicomponent Herbal Medicine in Healthy Chinese Volunteers.Clin Pharmacol Ther. 2018 Apr;103(4):692-702. doi: 10.1002/cpt.784. Epub 2017 Sep 19. Clin Pharmacol Ther. 2018. PMID: 28675423
-
An integrated metabolomics and pharmacokinetics strategy for multi-component drugs evaluation.Curr Drug Metab. 2010 Jan;11(1):105-14. doi: 10.2174/138920010791110926. Curr Drug Metab. 2010. PMID: 20302568 Review.
-
Preclinical pharmacokinetics: an approach towards safer and efficacious drugs.Curr Drug Metab. 2006 Feb;7(2):165-82. doi: 10.2174/138920006775541552. Curr Drug Metab. 2006. PMID: 16472106 Review.
-
Pharmacokinetics/pharmacometabolomics-pharmacodynamics reveals the synergistic mechanism of a multicomponent herbal formula, Baoyuan decoction against cardiac hypertrophy.Biomed Pharmacother. 2021 Jul;139:111665. doi: 10.1016/j.biopha.2021.111665. Epub 2021 May 7. Biomed Pharmacother. 2021. PMID: 34243607
-
polyPK: an R package for pharmacokinetic analysis of multi-component drugs using a metabolomics approach.Bioinformatics. 2018 May 15;34(10):1792-1794. doi: 10.1093/bioinformatics/btx834. Bioinformatics. 2018. PMID: 29293946 Free PMC article.
Cited by
-
Metabolic Changes of Maternal Uterine Fluid, Uterus, and Plasma during the Peri-implantation Period of Early Pregnancy in Mice.Reprod Sci. 2020 Feb;27(2):488-502. doi: 10.1007/s43032-019-00040-5. Epub 2020 Jan 6. Reprod Sci. 2020. PMID: 32046443
-
Pharmacokinetics and Metabolomic Profiling of Metformin and Andrographis paniculata: A Protocol for a Crossover Randomised Controlled Trial.J Clin Med. 2022 Jul 6;11(14):3931. doi: 10.3390/jcm11143931. J Clin Med. 2022. PMID: 35887695 Free PMC article.
-
Polyphenolic Nutraceuticals to Combat Oxidative Stress Through Microbiota Modulation.Front Pharmacol. 2019 May 3;10:492. doi: 10.3389/fphar.2019.00492. eCollection 2019. Front Pharmacol. 2019. PMID: 31130865 Free PMC article.
-
Hydroxylation Metabolisms of Crassicauline A in Rats Under Toxic Dose.Eur J Drug Metab Pharmacokinet. 2017 Oct;42(5):857-869. doi: 10.1007/s13318-017-0408-z. Eur J Drug Metab Pharmacokinet. 2017. PMID: 28276004
-
Recent highlights of metabolomics in chinese medicine syndrome research.Evid Based Complement Alternat Med. 2013;2013:402159. doi: 10.1155/2013/402159. Epub 2013 Nov 4. Evid Based Complement Alternat Med. 2013. PMID: 24302964 Free PMC article. Review.
References
-
- Blaylock RL. New developments in phytoprevention and treatment of cancer. The Journal of the American Nutraceutical Association. 1999;2:19–29.
-
- Hollander JM, Mechanick JI. Complementary and alternative medicine and the management of the metabolic syndrome. Journal of the American Dietetic Association. 2008;108(3):495–509. - PubMed
-
- Chan K. Progress in traditional Chinese medicine. Trends in Pharmacological Sciences. 1995;16(6):182–187. - PubMed
-
- Plumb GW, Garcia-Conesa MT, Kroon PA, Rhodes M, Ridley S, Williamson G. Metabolism of chlorogenic acid by human plasma, liver, intestine and gut microflora. Journal of the Science of Food and Agriculture. 1999;79(3):390–392.
-
- He SM, Chan E, Zhou SF. ADME properties of herbal medicines in humans: evidence, challenges andstrategies. Current Pharmaceutical Design. 2011;17(4):357–407. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

