The deoxyhypusine synthase mutant dys1-1 reveals the association of eIF5A and Asc1 with cell wall integrity
- PMID: 23573236
- PMCID: PMC3613415
- DOI: 10.1371/journal.pone.0060140
The deoxyhypusine synthase mutant dys1-1 reveals the association of eIF5A and Asc1 with cell wall integrity
Abstract
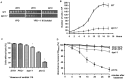
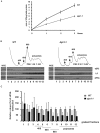
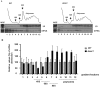
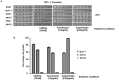
The putative eukaryotic translation initiation factor 5A (eIF5A) is a highly conserved protein among archaea and eukaryotes that has recently been implicated in the elongation step of translation. eIF5A undergoes an essential and conserved posttranslational modification at a specific lysine to generate the residue hypusine. The enzymes deoxyhypusine synthase (Dys1) and deoxyhypusine hydroxylase (Lia1) catalyze this two-step modification process. Although several Saccharomyces cerevisiae eIF5A mutants have importantly contributed to the study of eIF5A function, no conditional mutant of Dys1 has been described so far. In this study, we generated and characterized the dys1-1 mutant, which showed a strong depletion of mutated Dys1 protein, resulting in more than 2-fold decrease in hypusine levels relative to the wild type. The dys1-1 mutant demonstrated a defect in total protein synthesis, a defect in polysome profile indicative of a translation elongation defect and a reduced association of eIF5A with polysomes. The growth phenotype of dys1-1 mutant is severe, growing only in the presence of 1 M sorbitol, an osmotic stabilizer. Although this phenotype is characteristic of Pkc1 cell wall integrity mutants, the sorbitol requirement from dys1-1 is not associated with cell lysis. We observed that the dys1-1 genetically interacts with the sole yeast protein kinase C (Pkc1) and Asc1, a component of the 40S ribosomal subunit. The dys1-1 mutant was synthetically lethal in combination with asc1Δ and overexpression of TIF51A (eIF5A) or DYS1 is toxic for an asc1Δ strain. Moreover, eIF5A is more associated with translating ribosomes in the absence of Asc1 in the cell. Finally, analysis of the sensitivity to cell wall-perturbing compounds revealed a more similar behavior of the dys1-1 and asc1Δ mutants in comparison with the pkc1Δ mutant. These data suggest a correlated role for eIF5A and Asc1 in coordinating the translational control of a subset of mRNAs associated with cell integrity.
Conflict of interest statement
Figures



Similar articles
-
Hypusine-containing protein eIF5A promotes translation elongation.Nature. 2009 May 7;459(7243):118-21. doi: 10.1038/nature08034. Nature. 2009. PMID: 19424157 Free PMC article.
-
Tandem affinity purification revealed the hypusine-dependent binding of eukaryotic initiation factor 5A to the translating 80S ribosomal complex.J Cell Biochem. 2006 Feb 15;97(3):583-98. doi: 10.1002/jcb.20658. J Cell Biochem. 2006. PMID: 16215987
-
Mapping eIF5A binding sites for Dys1 and Lia1: in vivo evidence for regulation of eIF5A hypusination.FEBS Lett. 2003 Dec 18;555(3):464-8. doi: 10.1016/s0014-5793(03)01305-x. FEBS Lett. 2003. PMID: 14675757
-
The post-translational synthesis of a polyamine-derived amino acid, hypusine, in the eukaryotic translation initiation factor 5A (eIF5A).J Biochem. 2006 Feb;139(2):161-9. doi: 10.1093/jb/mvj034. J Biochem. 2006. PMID: 16452303 Free PMC article. Review.
-
Post-translational formation of hypusine in eIF5A: implications in human neurodevelopment.Amino Acids. 2022 Apr;54(4):485-499. doi: 10.1007/s00726-021-03023-6. Epub 2021 Jul 17. Amino Acids. 2022. PMID: 34273022 Free PMC article. Review.
Cited by
-
Differential gene expression in Aspergillus fumigatus induced by human platelets in vitro.Int J Med Microbiol. 2015 May;305(3):327-38. doi: 10.1016/j.ijmm.2015.01.002. Epub 2015 Jan 17. Int J Med Microbiol. 2015. PMID: 25661519 Free PMC article.
-
Fertility and polarized cell growth depends on eIF5A for translation of polyproline-rich formins in Saccharomyces cerevisiae.Genetics. 2014 Aug;197(4):1191-200. doi: 10.1534/genetics.114.166926. Epub 2014 Jun 11. Genetics. 2014. PMID: 24923804 Free PMC article.
-
Validation of Plasmodium falciparum deoxyhypusine synthase as an antimalarial target.PeerJ. 2019 Apr 17;7:e6713. doi: 10.7717/peerj.6713. eCollection 2019. PeerJ. 2019. PMID: 31024761 Free PMC article.
-
Evidence for a Negative Cooperativity between eIF5A and eEF2 on Binding to the Ribosome.PLoS One. 2016 Apr 26;11(4):e0154205. doi: 10.1371/journal.pone.0154205. eCollection 2016. PLoS One. 2016. PMID: 27115996 Free PMC article.
-
Roles of Rack1 Proteins in Fungal Pathogenesis.Biomed Res Int. 2016;2016:4130376. doi: 10.1155/2016/4130376. Epub 2016 Aug 30. Biomed Res Int. 2016. PMID: 27656651 Free PMC article. Review.
References
-
- Acker MG, Kolitz SE, Mitchell SF, Nanda JS, Lorsch JR (2007) Reconstitution of yeast translation initiation. Methods Enzymol 430: 111–145. - PubMed
-
- Kang HA, Hershey JW (1994) Effect of initiation factor eIF-5A depletion on protein synthesis and proliferation of Saccharomyces cerevisiae. J Biol Chem 269: 3934–3940. - PubMed
-
- Park MH, Lee YB, Joe YA (1997) Hypusine is essential for eukaryotic cell proliferation. Biological Signals 6: 115–123. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

