Effects of mechano-electric feedback on scroll wave stability in human ventricular fibrillation
- PMID: 23573245
- PMCID: PMC3616032
- DOI: 10.1371/journal.pone.0060287
Effects of mechano-electric feedback on scroll wave stability in human ventricular fibrillation
Abstract
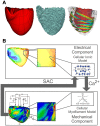
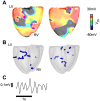
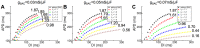
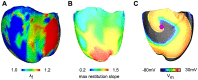
Recruitment of stretch-activated channels, one of the mechanisms of mechano-electric feedback, has been shown to influence the stability of scroll waves, the waves that underlie reentrant arrhythmias. However, a comprehensive study to examine the effects of recruitment of stretch-activated channels with different reversal potentials and conductances on scroll wave stability has not been undertaken; the mechanisms by which stretch-activated channel opening alters scroll wave stability are also not well understood. The goals of this study were to test the hypothesis that recruitment of stretch-activated channels affects scroll wave stability differently depending on stretch-activated channel reversal potential and channel conductance, and to uncover the relevant mechanisms underlying the observed behaviors. We developed a strongly-coupled model of human ventricular electromechanics that incorporated human ventricular geometry and fiber and sheet orientation reconstructed from MR and diffusion tensor MR images. Since a wide variety of reversal potentials and channel conductances have been reported for stretch-activated channels, two reversal potentials, -60 mV and -10 mV, and a range of channel conductances (0 to 0.07 mS/µF) were implemented. Opening of stretch-activated channels with a reversal potential of -60 mV diminished scroll wave breakup for all values of conductances by flattening heterogeneously the action potential duration restitution curve. Opening of stretch-activated channels with a reversal potential of -10 mV inhibited partially scroll wave breakup at low conductance values (from 0.02 to 0.04 mS/µF) by flattening heterogeneously the conduction velocity restitution relation. For large conductance values (>0.05 mS/µF), recruitment of stretch-activated channels with a reversal potential of -10 mV did not reduce the likelihood of scroll wave breakup because Na channel inactivation in regions of large stretch led to conduction block, which counteracted the increased scroll wave stability due to an overall flatter conduction velocity restitution.
Conflict of interest statement
Figures
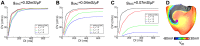
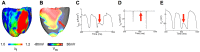
References
-
- Kohl P, Hunter P, Noble D (1999) Stretch-induced changes in heart rate and rhythm: clinical observations, experiments and mathematical models. Prog Biophys Mol Biol 71: 91–138. - PubMed
-
- Kohl P, Ravens U (2003) Cardiac mechano-electric feedback: past, present, and prospect. Prog Biophys Mol Biol 82: 3–9. - PubMed
-
- Miragoli M, Gaudesius G, Rohr S (2006) Electrotonic modulation of cardiac impulse conduction by myofibroblasts. Circ Res 98: 801–810. - PubMed
-
- Hu H, Sachs F (1997) Stretch-activated ion channels in the heart. J Mol Cell Cardiol 29: 1511–1523. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

