Invasion of primary glioma- and cell line-derived spheroids implanted into corticostriatal slice cultures
- PMID: 23573302
- PMCID: PMC3606845
Invasion of primary glioma- and cell line-derived spheroids implanted into corticostriatal slice cultures
Abstract
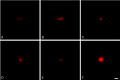
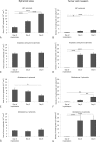
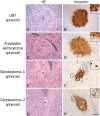
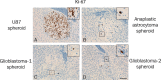
Gliomas are highly invasive tumors and the pronounced invasive features of gliomas prevent radical surgical resection. In the search for new therapeutics targeting invasive glioma cells, in vivo-like in vitro models are of great interest. We developed and evaluated an in vivo-like in vitro model preserving the invasive features and stem cell features of glioma cells. Fluorescently labelled primary glioma spheroids and U87MG cell line-derived spheroids were implanted into organotypic rat corticostriatal slice cultures and the invasion was followed over time by confocal microscopy. The invasion was validated immunohistochemically with paraffin sections using a human-specific vimentin antibody. Moreover, the preservation of immature stem cell features was evaluated immunohistochemically using the stem cell markers CD133, Sox2, Bmi-1 and nestin. The confocal and immunohistochemical results showed that the primary glioma spheroid area was constant or decreasing after implantation, with a clear increase in the number of invading cells over time. In contrast, the U87MG spheroid area increased after implantation, with no convincing tumor cell invasion. High levels of Bmi-1 and nestin were found in all spheroids, whereas high levels of Sox2 and low to moderate levels of CD133 were only found in the primary spheroids. In conclusion, the invasion of gliomas is preserved using primary glioma spheroids. Some stem cell features are preserved as well, making this model useful in drug development elucidating both invasion and cancer stemness at the early in vitro level.
Keywords: Glioma; invasion; organotypic slice cultures; spheroids.
Figures



Similar articles
-
CD133+ and nestin+ tumor-initiating cells dominate in N29 and N32 experimental gliomas.Int J Cancer. 2009 Jul 1;125(1):15-22. doi: 10.1002/ijc.24306. Int J Cancer. 2009. PMID: 19291792
-
[Correlative study of distribution of brain tumor stem cell with micro-vascular system].Zhonghua Yi Xue Za Zhi. 2010 Feb 2;90(5):305-9. Zhonghua Yi Xue Za Zhi. 2010. PMID: 20368050 Chinese.
-
Establishment and Characterization of a Tumor Stem Cell-Based Glioblastoma Invasion Model.PLoS One. 2016 Jul 25;11(7):e0159746. doi: 10.1371/journal.pone.0159746. eCollection 2016. PLoS One. 2016. PMID: 27454178 Free PMC article.
-
What is the clinical value of cancer stem cell markers in gliomas?Int J Clin Exp Pathol. 2013;6(3):334-48. Epub 2013 Feb 15. Int J Clin Exp Pathol. 2013. PMID: 23412423 Free PMC article. Review.
-
Stem cell markers in gliomas.Neurochem Res. 2008 Dec;33(12):2407-15. doi: 10.1007/s11064-008-9723-8. Epub 2008 May 21. Neurochem Res. 2008. PMID: 18493853 Review.
Cited by
-
Development of a 3D Tumor Spheroid Model from the Patient's Glioblastoma Cells and Its Study by Metabolic Fluorescence Lifetime Imaging.Sovrem Tekhnologii Med. 2023;15(2):28-38. doi: 10.17691/stm2023.15.2.03. Epub 2023 Mar 29. Sovrem Tekhnologii Med. 2023. PMID: 37389023 Free PMC article.
-
Shift of microRNA profile upon glioma cell migration using patient-derived spheroids and serum-free conditions.J Neurooncol. 2017 Mar;132(1):45-54. doi: 10.1007/s11060-016-2356-x. Epub 2017 Jan 13. J Neurooncol. 2017. PMID: 28091986 Free PMC article.
-
Microglia and astrocytes attenuate the replication of the oncolytic vaccinia virus LIVP 1.1.1 in murine GL261 gliomas by acting as vaccinia virus traps.J Transl Med. 2015 Jul 7;13:216. doi: 10.1186/s12967-015-0586-x. J Transl Med. 2015. PMID: 26149494 Free PMC article.
-
Gene Expression Profile of 3D Spheroids in Comparison with 2D Cell Cultures and Tissue Strains of Diffuse High-Grade Gliomas.Bull Exp Biol Med. 2023 Aug;175(4):576-584. doi: 10.1007/s10517-023-05906-y. Epub 2023 Sep 29. Bull Exp Biol Med. 2023. PMID: 37770789
-
Microglia induce an interferon-stimulated gene expression profile in glioblastoma and increase glioblastoma resistance to temozolomide.Neuropathol Appl Neurobiol. 2024 Dec;50(6):e13016. doi: 10.1111/nan.13016. Neuropathol Appl Neurobiol. 2024. PMID: 39558550 Free PMC article.
References
-
- Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, Ludwin SK, Allgeier A, Fisher B, Belanger K, Hau P, Brandes AA, Gijtenbeek J, Marosi C, Vecht CJ, Mokhtari K, Wesseling P, Villa S, Eisenhauer E, Gorlia T, Weller M, Lacombe D, Cairncross JG, Mirimanoff RO European Organisation for Research and Treatment of Cancer Brain Tumour and Radiation Oncology Groups; National Cancer Institute of Canada Clinical Trials Group. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009;10:459–466. - PubMed
-
- Keunen O, Johansson M, Oudin A, Sanzey M, Rahim SA, Fack F, Thorsen F, Taxt T, Bartos M, Jirik R, Miletic H, Wang J, Stieber D, Stuhr L, Moen I, Rygh CB, Bjerkvig R, Niclou SP. Anti-VEGF treatment reduces blood supply and increases tumor cell invasion in glioblastoma. Proc Natl Acad Sci U S A. 2011;108:3749–3754. - PMC - PubMed
-
- Bjerkvig R, Tonnesen A, Laerum OD, Backlund EO. Multicellular tumor spheroids from human gliomas maintained in organ culture. J Neurosurg. 1990;72:463–475. - PubMed
-
- De Witt Hamer PC, Jonker A, Leenstra S, Ruijter JM, Van Noorden CJ. Quantification of viability in organotypic multicellular spheroids of human malignant glioma using lactate dehydrogenase activity: a rapid and reliable automated assay. J Histochem Cytochem. 2005;53:23–34. - PubMed
-
- Kaaijk P, Troost D, Das PK, Leenstra S, Bosch DA. Long-term culture of organotypic multicellular glioma spheroids: a good culture model for studying gliomas. Neuropathol Appl Neurobiol. 1995;21:386–391. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
