Progesterone receptors induce FOXO1-dependent senescence in ovarian cancer cells
- PMID: 23574718
- PMCID: PMC3674071
- DOI: 10.4161/cc.24550
Progesterone receptors induce FOXO1-dependent senescence in ovarian cancer cells
Abstract
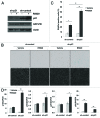
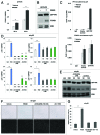
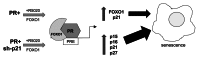
Loss of nuclear progesterone receptors (PR) and low circulating progesterone levels are associated with increased ovarian cancer (OC) risk. However, PR are abundantly expressed in a significant percentage of serous and endometrioid ovarian tumors; patients with PR+ tumors typically experience longer progression-free survival relative to those with PR-null tumors. The molecular mechanisms of these protective effects are poorly understood. To study PR action in OC in the absence of added estrogen (i.e., needed to induce robust PR expression), we created ES-2 OC cells stably expressing vector control or GFP-tagged PR-B (GFP-PR). Progestin (R5020) stimulation of ES-2 cells stably expressing GFP-PR induced cellular senescence characterized by altered cellular morphology, prolonged survival, senescence-associated β-galactosidase activity, G1 cell cycle arrest and upregulation of the cell cycle inhibitor, p21, as well as the Forkhead-box transcription factor, FOXO1; these results repeated in unmodified ER+/PR+ PEO4 OC cells. PR-B and FOXO1 were detected within the same PRE-containing regions of the p21 upstream promoter. Knockdown of p21 resulted in molecular compensation via FOXO1-dependent upregulation of numerous FOXO1 target genes (p15, p16, p27) and an increased rate of senescence. Inhibition of FOXO1 (with AS1842856) or stable FOXO1 knockdown inhibited progestin-induced p21 expression and blocked progestin-induced senescence. Overall, these findings support a role for PR as a tumor suppressor in OC cells, which exhibits inhibitory effects by inducing FOXO1-dependent cellular senescence. Clinical "priming" of the PR-FOXO1-p21 signaling pathway using PR agonists may provide a useful strategy to induce irreversible cell cycle arrest and thereby sensitize OC cells to existing chemotherapies as part of combination "two-step" therapies.
Keywords: AS1842856; FOXO1; breast cancer; forkhead transcription factor; ovarian cancer; p21; progesterone receptor; progestin; senescence.
Figures
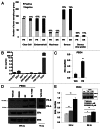
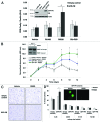
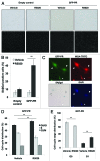
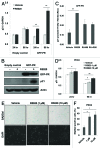

Comment in
-
Progestins: pro-senescence therapy for ovarian cancer?Cell Cycle. 2013 Jun 1;12(11):1662-3. doi: 10.4161/cc.25072. Epub 2013 May 17. Cell Cycle. 2013. PMID: 23708445 Free PMC article. No abstract available.
-
Progesterone and FOXO1 signaling: harnessing cellular senescence for the treatment of ovarian cancer.Cell Cycle. 2013 Jun 1;12(11):1660-1. doi: 10.4161/cc.25070. Epub 2013 May 17. Cell Cycle. 2013. PMID: 23708447 Free PMC article. No abstract available.
Similar articles
-
Active FOXO1 Is a Key Determinant of Isoform-Specific Progesterone Receptor Transactivation and Senescence Programming.Mol Cancer Res. 2016 Feb;14(2):141-62. doi: 10.1158/1541-7786.MCR-15-0431. Epub 2015 Nov 17. Mol Cancer Res. 2016. PMID: 26577046 Free PMC article.
-
Forkhead transcription factor FOXO1 is a direct target of progestin to inhibit endometrial epithelial cell growth.Clin Cancer Res. 2011 Feb 1;17(3):525-37. doi: 10.1158/1078-0432.CCR-10-1287. Epub 2010 Dec 3. Clin Cancer Res. 2011. PMID: 21131554
-
Curcumin induces apoptosis in pancreatic cancer cells through the induction of forkhead box O1 and inhibition of the PI3K/Akt pathway.Mol Med Rep. 2015 Oct;12(4):5415-22. doi: 10.3892/mmr.2015.4060. Epub 2015 Jul 8. Mol Med Rep. 2015. PMID: 26166196
-
FOXO1, a tiny protein with intricate interactions: Promising therapeutic candidate in lung cancer.Biomed Pharmacother. 2023 Dec 31;169:115900. doi: 10.1016/j.biopha.2023.115900. Epub 2023 Nov 18. Biomed Pharmacother. 2023. PMID: 37981461 Review.
-
Acupuncture and cellular mechanisms: the role of foxo transcription factors in autophagy and senescence.J Pak Med Assoc. 2024 Dec;74(12):2150-2154. doi: 10.47391/JPMA.10423. J Pak Med Assoc. 2024. PMID: 39658986 Review.
Cited by
-
Progestins: pro-senescence therapy for ovarian cancer?Cell Cycle. 2013 Jun 1;12(11):1662-3. doi: 10.4161/cc.25072. Epub 2013 May 17. Cell Cycle. 2013. PMID: 23708445 Free PMC article. No abstract available.
-
Mechanisms and significance of therapy-induced and spontaneous senescence of cancer cells.Cell Mol Life Sci. 2020 Jan;77(2):213-229. doi: 10.1007/s00018-019-03261-8. Epub 2019 Aug 14. Cell Mol Life Sci. 2020. PMID: 31414165 Free PMC article. Review.
-
Reevaluating the Role of Progesterone in Ovarian Cancer: Is Progesterone Always Protective?Endocr Rev. 2023 Nov 9;44(6):1029-1046. doi: 10.1210/endrev/bnad018. Endocr Rev. 2023. PMID: 37261958 Free PMC article.
-
Deletion of PDK1 causes cardiac sodium current reduction in mice.PLoS One. 2015 Mar 17;10(3):e0122436. doi: 10.1371/journal.pone.0122436. eCollection 2015. PLoS One. 2015. PMID: 25781322 Free PMC article.
-
Selective inhibition of progesterone receptor in osteochondral progenitor cells, but not in mature chondrocytes, modulated subchondral bone structures.Bone. 2020 Mar;132:115196. doi: 10.1016/j.bone.2019.115196. Epub 2019 Dec 19. Bone. 2020. PMID: 31863959 Free PMC article.
References
-
- Atlanta: American Cancer Society Cancer Facts & Figures. 2012.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
