A herpes simplex virus scaffold peptide that binds the portal vertex inhibits early steps in viral replication
- PMID: 23576509
- PMCID: PMC3676109
- DOI: 10.1128/JVI.00421-13
A herpes simplex virus scaffold peptide that binds the portal vertex inhibits early steps in viral replication
Abstract
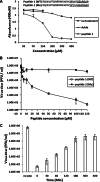
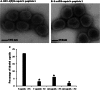
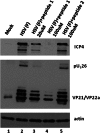
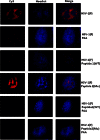
Previous experiments identified a 12-amino-acid (aa) peptide that was sufficient to interact with the herpes simplex virus 1 (HSV-1) portal protein and was necessary to incorporate the portal into capsids. In the present study, cells were treated at various times postinfection with peptides consisting of a portion of the Drosophila antennapedia protein, previously shown to enter cells efficiently, fused to either wild-type HSV-1 scaffold peptide (YPYYPGEARGAP) or a control peptide that contained changes at positions 4 and 5. These 4-tyrosine and 5-proline residues are highly conserved in herpesvirus scaffold proteins and were previously shown to be critical for the portal interaction. Treatment early in infection with subtoxic levels of wild-type peptide reduced viral infectivity by over 1,000-fold, while the mutant peptide had little effect on viral yields. In cells infected for 3 h in the presence of wild-type peptide, capsids were observed to transit to the nuclear rim normally, as viewed by fluorescence microscopy. However, observation by electron microscopy in thin sections revealed an aberrant and significant increase of DNA-containing capsids compared to infected cells treated with the mutant peptide. Early treatment with peptide also prevented formation of viral DNA replication compartments. These data suggest that the antiviral peptide stabilizes capsids early in infection, causing retention of DNA within them, and that this activity correlates with peptide binding to the portal protein. The data are consistent with the hypothesis that the portal vertex is the conduit through which DNA is ejected to initiate infection.
Figures





Similar articles
-
Proline and tyrosine residues in scaffold proteins of herpes simplex virus 1 critical to the interaction with portal protein and its incorporation into capsids.J Virol. 2009 Aug;83(16):8076-81. doi: 10.1128/JVI.00655-09. Epub 2009 May 27. J Virol. 2009. PMID: 19474093 Free PMC article.
-
Role of the Herpes Simplex Virus CVSC Proteins at the Capsid Portal Vertex.J Virol. 2020 Nov 23;94(24):e01534-20. doi: 10.1128/JVI.01534-20. Print 2020 Nov 23. J Virol. 2020. PMID: 32967953 Free PMC article.
-
Domain within herpes simplex virus 1 scaffold proteins required for interaction with portal protein in infected cells and incorporation of the portal vertex into capsids.J Virol. 2008 May;82(10):5021-30. doi: 10.1128/JVI.00150-08. Epub 2008 Mar 12. J Virol. 2008. PMID: 18337579 Free PMC article.
-
Tryptophan residues in the portal protein of herpes simplex virus 1 critical to the interaction with scaffold proteins and incorporation of the portal into capsids.J Virol. 2009 Nov;83(22):11726-33. doi: 10.1128/JVI.01463-09. Epub 2009 Sep 9. J Virol. 2009. PMID: 19740984 Free PMC article.
-
Herpesvirus capsid assembly: insights from structural analysis.Curr Opin Virol. 2011 Aug;1(2):142-9. doi: 10.1016/j.coviro.2011.06.003. Curr Opin Virol. 2011. PMID: 21927635 Free PMC article. Review.
Cited by
-
Cryo-electron microscopy structures of capsids and in situ portals of DNA-devoid capsids of human cytomegalovirus.Nat Commun. 2023 Apr 11;14(1):2025. doi: 10.1038/s41467-023-37779-0. Nat Commun. 2023. PMID: 37041152 Free PMC article.
-
Terminase Large Subunit Provides a New Drug Target for Herpesvirus Treatment.Viruses. 2019 Mar 5;11(3):219. doi: 10.3390/v11030219. Viruses. 2019. PMID: 30841485 Free PMC article. Review.
References
-
- Elion GB. 1982. Mechanism of action and selectivity of acyclovir. Am. J. Med. 73:7–13 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

