Synthetic microRNA-mediated downregulation of Nogo-A in transgenic rats reveals its role as regulator of synaptic plasticity and cognitive function
- PMID: 23576723
- PMCID: PMC3631667
- DOI: 10.1073/pnas.1217665110
Synthetic microRNA-mediated downregulation of Nogo-A in transgenic rats reveals its role as regulator of synaptic plasticity and cognitive function
Abstract
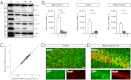
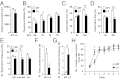
We have generated a transgenic rat model using RNAi and used it to study the role of the membrane protein Nogo-A in synaptic plasticity and cognition. The membrane protein Nogo-A is expressed in CNS oligodendrocytes and subpopulations of neurons, and it is known to suppress neurite growth and regeneration. The constitutively expressed polymerase II-driven transgene was composed of a microRNA-targeting Nogo-A placed into an intron preceding the coding sequence for EGFP, thus quantitatively labeling cells according to intracellular microRNA expression. The transgenic microRNA in vivo efficiently reduced the concentration of Nogo-A mRNA and protein preferentially in neurons. The resulting significant increase in long-term potentiation in both hippocampus and motor cortex indicates a repressor function of Nogo-A in synaptic plasticity. The transgenic rats exhibited prominent schizophrenia-like behavioral phenotypes, such as perseveration, disrupted prepulse inhibition, and strong withdrawal from social interactions. This fast and efficient microRNA-mediated knockdown provides a way to silence gene expression in vivo in transgenic rats and shows a role of Nogo-A in regulating higher cognitive brain functions.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Neutralization of Nogo-A enhances synaptic plasticity in the rodent motor cortex and improves motor learning in vivo.J Neurosci. 2014 Jun 25;34(26):8685-98. doi: 10.1523/JNEUROSCI.3817-13.2014. J Neurosci. 2014. PMID: 24966370 Free PMC article.
-
NogoA restricts synaptic plasticity in the adult hippocampus on a fast time scale.Proc Natl Acad Sci U S A. 2011 Feb 8;108(6):2569-74. doi: 10.1073/pnas.1013322108. Epub 2011 Jan 24. Proc Natl Acad Sci U S A. 2011. PMID: 21262805 Free PMC article.
-
The sphingolipid receptor S1PR2 is a receptor for Nogo-a repressing synaptic plasticity.PLoS Biol. 2014 Jan;12(1):e1001763. doi: 10.1371/journal.pbio.1001763. Epub 2014 Jan 14. PLoS Biol. 2014. PMID: 24453941 Free PMC article.
-
The role of Nogo-A in axonal plasticity, regrowth and repair.Cell Tissue Res. 2012 Jul;349(1):97-104. doi: 10.1007/s00441-012-1432-6. Epub 2012 May 17. Cell Tissue Res. 2012. PMID: 22588543 Review.
-
The Nogo receptor, its ligands and axonal regeneration in the spinal cord; a review.J Neurocytol. 2002 Feb;31(2):93-120. doi: 10.1023/a:1023941421781. J Neurocytol. 2002. PMID: 12815233 Review.
Cited by
-
Acquisition of a High-precision Skilled Forelimb Reaching Task in Rats.J Vis Exp. 2015 Jun 22;(100):e53010. doi: 10.3791/53010. J Vis Exp. 2015. PMID: 26131653 Free PMC article.
-
The Neurite Outgrowth Inhibitory Nogo-A-Δ20 Region Is an Intrinsically Disordered Segment Harbouring Three Stretches with Helical Propensity.PLoS One. 2016 Sep 9;11(9):e0161813. doi: 10.1371/journal.pone.0161813. eCollection 2016. PLoS One. 2016. PMID: 27611089 Free PMC article.
-
Urokinase-type Plasminogen Activator Induces Neurorepair in the Ischemic Brain.J Neurol Exp Neurosci. 2018;4(2):24-29. doi: 10.17756/jnen.2018-039. Epub 2018 Sep 26. J Neurol Exp Neurosci. 2018. PMID: 30417161 Free PMC article.
-
The Implication of Reticulons (RTNs) in Neurodegenerative Diseases: From Molecular Mechanisms to Potential Diagnostic and Therapeutic Approaches.Int J Mol Sci. 2021 Apr 28;22(9):4630. doi: 10.3390/ijms22094630. Int J Mol Sci. 2021. PMID: 33924890 Free PMC article. Review.
-
Reduced expression of nogo-a leads to motivational deficits in rats.Front Behav Neurosci. 2014 Jan 22;8:10. doi: 10.3389/fnbeh.2014.00010. eCollection 2014. Front Behav Neurosci. 2014. PMID: 24478657 Free PMC article.
References
-
- Glaser S, Anastassiadis K, Stewart AF. Current issues in mouse genome engineering. Nat Genet. 2005;37(11):1187–1193. - PubMed
-
- Hemann MT, et al. An epi-allelic series of p53 hypomorphs created by stable RNAi produces distinct tumor phenotypes in vivo. Nat Genet. 2003;33(3):396–400. - PubMed
-
- Aitman TJ, et al. Progress and prospects in rat genetics: A community view. Nat Genet. 2008;40(5):516–522. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

