Activation of NF-kappa B signaling promotes growth of prostate cancer cells in bone
- PMID: 23577181
- PMCID: PMC3618119
- DOI: 10.1371/journal.pone.0060983
Activation of NF-kappa B signaling promotes growth of prostate cancer cells in bone
Abstract
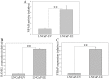
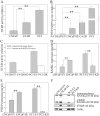
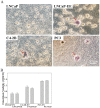
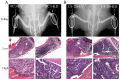
Patients with advanced prostate cancer almost invariably develop osseous metastasis. Although many studies indicate that the activation of NF-κB signaling appears to be correlated with advanced cancer and promotes tumor metastasis by influencing tumor cell migration and angiogenesis, the influence of altered NF-κB signaling in prostate cancer cells within boney metastatic lesions is not clearly understood. While C4-2B and PC3 prostate cancer cells grow well in the bone, LNCaP cells are difficult to grow in murine bone following intraskeletal injection. Our studies show that when compared to LNCaP, NF-κB activity is significantly higher in C4-2B and PC3, and that the activation of NF-κB signaling in prostate cancer cells resulted in the increased expression of the osteoclast inducing genes PTHrP and RANKL. Further, conditioned medium derived from NF-κB activated LNCaP cells induce osteoclast differentiation. In addition, inactivation of NF-κB signaling in prostate cancer cells inhibited tumor formation in the bone, both in the osteolytic PC3 and osteoblastic/osteoclastic mixed C4-2B cells; while the activation of NF-κB signaling in LNCaP cells promoted tumor establishment and proliferation in the bone. The activation of NF-κB in LNCaP cells resulted in the formation of an osteoblastic/osteoclastic mixed tumor with increased osteoclasts surrounding the new formed bone, similar to metastases commonly seen in patients with prostate cancer. These results indicate that osteoclastic reaction is required even in the osteoblastic cancer cells and the activation of NF-κB signaling in prostate cancer cells increases osteoclastogenesis by up-regulating osteoclastogenic genes, thereby contributing to bone metastatic formation.
Conflict of interest statement
Figures



References
-
- Mundy GR (2002) Metastasis to bone: causes, consequences and therapeutic opportunities. Nat Rev Cancer 2: 584–593. - PubMed
-
- Cheville JC, Tindall D, Boelter C, Jenkins R, Lohse CM, et al. (2002) Metastatic prostate carcinoma to bone: clinical and pathologic features associated with cancer-specific survival. Cancer 95: 1028–1036. - PubMed
-
- Pound CR, Partin AW, Eisenberger MA, Chan DW, Pearson JD, et al. (1999) Natural history of progression after PSA elevation following radical prostatectomy. Jama 281: 1591–1597. - PubMed
-
- Roodman GD (2004) Mechanisms of bone metastasis. N Engl J Med 350: 1655–1664. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

