Wedelolactone, a naturally occurring coumestan, enhances interferon-γ signaling through inhibiting STAT1 protein dephosphorylation
- PMID: 23580655
- PMCID: PMC3656297
- DOI: 10.1074/jbc.M112.442970
Wedelolactone, a naturally occurring coumestan, enhances interferon-γ signaling through inhibiting STAT1 protein dephosphorylation
Abstract
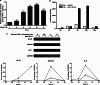
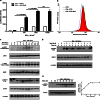
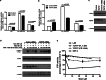
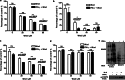
Signal transducers and activators of transcription 1 (STAT1) transduces signals from cytokines and growth factors, particularly IFN-γ, and regulates expression of genes involved in cell survival/death, proliferation, and migration. STAT1 is activated through phosphorylation on its tyrosine 701 by JAKs and is inactivated through dephosphorylation by tyrosine phosphatases. We discovered a natural compound, wedelolactone, that increased IFN-γ signaling by inhibiting STAT1 dephosphorylation and prolonging STAT1 activation through specific inhibition of T-cell protein tyrosine phosphatase (TCPTP), an important tyrosine phosphatase for STAT1 dephosphorylation. More interestingly, wedelolactone inhibited TCPTP through interaction with the C-terminal autoinhibition domain of TCPTP. We also found that wedelolactone synergized with IFN-γ to induce apoptosis of tumor cells. Our data suggest a new target for anticancer or antiproliferation drugs, a new mechanism to regulate PTPs specifically, and a new drug candidate for treating cancer or other proliferation disorders.
Keywords: Cytokines/Interferon; Phosphatase; STAT Transcription Factor; Tumor Suppressor Gene; Tumor Therapy.
Figures


Similar articles
-
Negative regulation of interferon-γ/STAT1 signaling through cell adhesion and cell density-dependent STAT1 dephosphorylation.Cell Signal. 2011 Aug;23(8):1404-12. doi: 10.1016/j.cellsig.2011.04.003. Epub 2011 Apr 13. Cell Signal. 2011. PMID: 21511030
-
Luteolin sensitizes the antiproliferative effect of interferon α/β by activation of Janus kinase/signal transducer and activator of transcription pathway signaling through protein kinase A-mediated inhibition of protein tyrosine phosphatase SHP-2 in cancer cells.Cell Signal. 2014 Mar;26(3):619-28. doi: 10.1016/j.cellsig.2013.11.039. Epub 2013 Dec 12. Cell Signal. 2014. PMID: 24333668
-
Activation of signal transducer and activator of transcription 1 (STAT1) is not sufficient for the induction of STAT1-dependent genes in endothelial cells. Comparison of interferon-gamma and oncostatin M.J Biol Chem. 2002 Mar 8;277(10):8012-21. doi: 10.1074/jbc.M107542200. Epub 2002 Jan 3. J Biol Chem. 2002. PMID: 11777927
-
Nuclear beta-arrestin1 functions as a scaffold for the dephosphorylation of STAT1 and moderates the antiviral activity of IFN-gamma.Mol Cell. 2008 Sep 5;31(5):695-707. doi: 10.1016/j.molcel.2008.06.017. Mol Cell. 2008. PMID: 18775329
-
Interferon-gamma inhibits interferon-alpha signalling in hepatic cells: evidence for the involvement of STAT1 induction and hyperexpression of STAT1 in chronic hepatitis C.Biochem J. 2004 Apr 1;379(Pt 1):199-208. doi: 10.1042/BJ20031495. Biochem J. 2004. PMID: 14690454 Free PMC article.
Cited by
-
Medicinal Plants from Near East for Cancer Therapy.Front Pharmacol. 2018 Jan 31;9:56. doi: 10.3389/fphar.2018.00056. eCollection 2018. Front Pharmacol. 2018. PMID: 29445343 Free PMC article. Review.
-
Protein tyrosine phosphatases as wardens of STAT signaling.JAKSTAT. 2014 Jan 1;3(1):e28087. doi: 10.4161/jkst.28087. Epub 2014 Feb 20. JAKSTAT. 2014. PMID: 24778927 Free PMC article. Review.
-
Exploring the recent trends in perturbing the cellular signaling pathways in cancer by natural products.Front Pharmacol. 2022 Sep 8;13:950109. doi: 10.3389/fphar.2022.950109. eCollection 2022. Front Pharmacol. 2022. PMID: 36160435 Free PMC article. Review.
-
Wedelolactone from Vietnamese Eclipta prostrata (L) L. Protected Zymosan-induced shock in Mice.Iran J Pharm Res. 2018 Spring;17(2):653-660. Iran J Pharm Res. 2018. PMID: 29881422 Free PMC article.
-
Interferon receptor signaling in malignancy: a network of cellular pathways defining biological outcomes.Mol Cancer Res. 2014 Dec;12(12):1691-703. doi: 10.1158/1541-7786.MCR-14-0450. Epub 2014 Sep 12. Mol Cancer Res. 2014. PMID: 25217450 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

