Circuits for grasping: spinal dI3 interneurons mediate cutaneous control of motor behavior
- PMID: 23583114
- PMCID: PMC4535710
- DOI: 10.1016/j.neuron.2013.02.007
Circuits for grasping: spinal dI3 interneurons mediate cutaneous control of motor behavior
Abstract
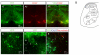
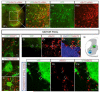
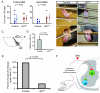
Accurate motor performance depends on the integration in spinal microcircuits of sensory feedback information. Hand grasp is a skilled motor behavior known to require cutaneous sensory feedback, but spinal microcircuits that process and relay this feedback to the motor system have not been defined. We sought to define classes of spinal interneurons involved in the cutaneous control of hand grasp in mice and to show that dI3 interneurons, a class of dorsal spinal interneurons marked by the expression of Isl1, convey input from low threshold cutaneous afferents to motoneurons. Mice in which the output of dI3 interneurons has been inactivated exhibit deficits in motor tasks that rely on cutaneous afferent input. Most strikingly, the ability to maintain grip strength in response to increasing load is lost following genetic silencing of dI3 interneuron output. Thus, spinal microcircuits that integrate cutaneous feedback crucial for paw grip rely on the intermediary role of dI3 interneurons.
Copyright © 2013 Elsevier Inc. All rights reserved.
Figures




References
-
- Abdala V, Manzano AS, Tulli MJ, Herrel A. The tendinous patterns in the palmar surface of the lizard manus: functional consequences for grasping ability. Anat. Rec. (Hoboken) 2009;292:842–853. - PubMed
-
- Akay T, Acharya HJ, Fouad K, Pearson KG. Behavioral and electromyographic characterization of mice lacking EphA4 receptors. J. Neurophysiol. 2006;96:642–651. - PubMed
-
- Alstermark B, Pettersson LG, Nishimura Y, Yoshino-Saito K, Tsuboi F, Takahashi M, Isa T. Motor command for precision grip in the macaque monkey can be mediated by spinal interneurons. J. Neurophysiol. 2011;106:122–126. - PubMed
-
- Alvarez FJ, Villalba RM, Zerda R, Schneider SP. Vesicular glutamate transporters in the spinal cord, with special reference to sensory primary afferent synapses. J. Comp. Neurol. 2004;472:257–280. - PubMed
-
- Amendola J, Verrier B, Roubertoux P, Durand J. Altered sensorimotor development in a transgenic mouse model of amyotrophic lateral sclerosis. Eur. J. Neurosci. 2004;20:2822–2826. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

