The translation of translational control by FMRP: therapeutic targets for FXS
- PMID: 23584741
- PMCID: PMC3999698
- DOI: 10.1038/nn.3379
The translation of translational control by FMRP: therapeutic targets for FXS
Abstract
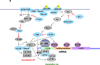
De novo protein synthesis is necessary for long-lasting modifications in synaptic strength and dendritic spine dynamics that underlie cognition. Fragile X syndrome (FXS), characterized by intellectual disability and autistic behaviors, holds promise for revealing the molecular basis for these long-term changes in neuronal function. Loss of function of the fragile X mental retardation protein (FMRP) results in defects in synaptic plasticity and cognition in many models of the disease. FMRP is a polyribosome-associated RNA-binding protein that regulates the synthesis of a set of plasticity-reated proteins by stalling ribosomal translocation on target mRNAs. The recent identification of mRNA targets of FMRP and its upstream regulators, and the use of small molecules to stall ribosomes in the absence of FMRP, have the potential to be translated into new therapeutic avenues for the treatment of FXS.
Figures


References
-
- Santoro MR, Bray SM, Warren ST. Molecular mechanisms of fragile X syndrome: a twenty-year perspective. Annu Rev Pathol. 2012;7:219–245. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

