Rpp25 is a major target of autoantibodies to the Th/To complex as measured by a novel chemiluminescent assay
- PMID: 23587095
- PMCID: PMC3672760
- DOI: 10.1186/ar4210
Rpp25 is a major target of autoantibodies to the Th/To complex as measured by a novel chemiluminescent assay
Abstract
Introduction: Autoantibodies to the Th/To antigen have been described in systemic sclerosis (SSc) and several proteins of the macromolecular Th/To complex have been reported to react with anti-Th/To antibodies. However, anti-Th/To has not been clinically utilized due to unavailability of commercial tests. The objective of the present study is to evaluate the newly developed ELISA and chemiluminescent immunoassay (CLIA) to measure autoantibodies to Rpp25 (a component of the Th/To complex) using immunoprecipitation (IP) as the reference method.
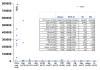
Methods: The first cohort consisted of 123 SSc patients including 7 anti-Th/To positive samples confirmed by IP. Additional seven anti-Th/To positive samples from non-SSc patients were also tested. For evaluation of the QUANTA Flash Rpp25 CLIA (research use only), 8 anti-Th/To IP positives, a cohort of 70 unselected SSc patients and sera from various disease controls (n = 357) and random healthy individuals (n = 10) were studied.
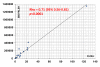
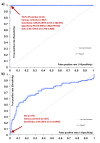
Results: Anti-Rpp25 antibodies determined by ELISA were found in 11/14 anti-Th/To IP positive but only in 1/156 (0.6%) negative samples resulting in a positive percent agreement of 78.6% (95% confidence interval [CI] 49.2, 95.3%) and a negative percent agreement of 99.4% (95% CI 96.4, 100.0%). To verify the results using a second method, 53 samples were tested by ELISA and CLIA for anti-Rpp25 reactivity and the results were highly correlated (rho = 0.71, 95% CI 0.56, 0.81; P < 0.0001). To define the cutoff of the CLIA, anti-Th/To IP positive and negative sera were tested using the anti-Rpp25 CLIA. At the cutoff selected by receiver operating characteristic (ROC) analysis 8/8 (100.0%) of the anti-Th/To positive sera but only 2/367 (0.5%) of the controls were positive for anti-Rpp25 antibodies. The positive and negative percent agreements were 100.0% (95% CI 63.1, 100.0%) and 99.5% (95% CI 98.0, 99.9%), respectively. In the disease cohorts 2/70 (2.9%) of the SSc patients were positive for anti-Rpp25 antibodies compared to 2/367 (0.5%) of the controls (P = 0.032). ROC analysis showed discrimination between SSc patients and controls with an area under the curve value of 0.732 (95% CI 0.655, 0.809).
Conclusion: Rpp25 is a major target of autoantibodies to the Th/To autoantigen complex. Further studies are needed to evaluate the clinical utility of the new assays.
Figures

Comment in
-
What autoantibody tests should become widely available to help scleroderma diagnosis and management?Arthritis Res Ther. 2013 Jul 8;15(4):116. doi: 10.1186/ar4241. Arthritis Res Ther. 2013. PMID: 23856077 Free PMC article.
References
-
- Fritzler MJ, Rattner JB, Luft LM, Edworthy SM, Casiano CA, Peebles C, Mahler M. Historical perspectives on the discovery and elucidation of autoantibodies to centromere proteins (CENP) and the emerging importance of antibodies to CENP-F. Autoimmun Rev. 2011;10:194–200. doi: 10.1016/j.autrev.2010.09.025. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

