The interaction of bortezomib with multidrug transporters: implications for therapeutic applications in advanced multiple myeloma and other neoplasias
- PMID: 23589314
- PMCID: PMC4414393
- DOI: 10.1007/s00280-013-2136-7
The interaction of bortezomib with multidrug transporters: implications for therapeutic applications in advanced multiple myeloma and other neoplasias
Abstract
Purpose: Bortezomib is an important agent in multiple myeloma treatment, but resistance in cell lines and patients has been described. The main mechanisms of resistance described in cancer fall into one of two categories, pharmacokinetic resistance (PK), e.g. over expression of drug efflux pumps and pharmacodynamic resistance, e.g. apoptosis resistance or altered survival pathways, where the agent reaches an appropriate concentration, but this fails to propagate an appropriate cell death response. Of the known pump mechanisms, P-glycoprotein (P-gp) is the best studied and considered to be the most important in contributing to general PK drug resistance. Resistance to bortezomib is multifactorial and there are conflicting indications that cellular overexpression of P-gp may contribute to resistance agent. Hence, better characterization of the interactions of this drug with classical resistance mechanisms should identify improved treatment applications.
Methods: Cell lines with different P-gp expression levels were used to determine the relationship between bortezomib and P-gp. Coculture system with stromal cells was used to determine the effect of the local microenvironment on the bortezomib-elacridar combination. To further assess P-gp function, intracellular accumulation of P-gp probe rhodamine-123 was utilised.
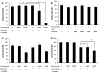
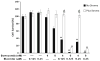
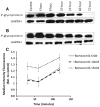
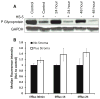
Results: In the present study, we show that bortezomib is a substrate for P-gp, but not for the other drug efflux transporters. Bortezomib activity is affected by P-gp expression and conversely, the expression of P-gp affect bortezomib's ability to act as a P-gp substrate. The local microenvironment did not alter the cellular response to bortezomib. We also demonstrate that bortezomib directly affects the expression and function of P-gp.
Conclusions: Our findings strongly support a role for P-gp in bortezomib resistance and, therefore, suggest that combination of a P-gp inhibitor and bortezomib in P-gp positive myeloma would be a reasonable treatment combination to extend efficacy of this important drug.
Conflict of interest statement
Figures


Similar articles
-
Tipifarnib and bortezomib are synergistic and overcome cell adhesion-mediated drug resistance in multiple myeloma and acute myeloid leukemia.Clin Cancer Res. 2006 Jan 15;12(2):591-9. doi: 10.1158/1078-0432.CCR-05-1792. Clin Cancer Res. 2006. PMID: 16428505
-
Suitable drug combination with bortezomib for multiple myeloma under stroma-free conditions and in contact with fibronectin or bone marrow stromal cells.Int J Hematol. 2014 Jun;99(6):726-36. doi: 10.1007/s12185-014-1573-3. Epub 2014 Apr 6. Int J Hematol. 2014. PMID: 24706190
-
Bortezomib restores stroma-mediated APO2L/TRAIL apoptosis resistance in multiple myeloma.Eur J Haematol. 2010 Mar;84(3):212-22. doi: 10.1111/j.1600-0609.2009.01381.x. Epub 2009 Nov 17. Eur J Haematol. 2010. PMID: 19922463
-
The role of P-glycoprotein in drug resistance in multiple myeloma.Leuk Lymphoma. 2015 Jan;56(1):26-33. doi: 10.3109/10428194.2014.907890. Epub 2014 Jun 5. Leuk Lymphoma. 2015. PMID: 24678978 Review.
-
Effects of bortezomib on the immune system: a focus on immune regulation.Biol Blood Marrow Transplant. 2013 Oct;19(10):1416-20. doi: 10.1016/j.bbmt.2013.05.011. Epub 2013 May 22. Biol Blood Marrow Transplant. 2013. PMID: 23707853 Review.
Cited by
-
Emerging Therapeutic Strategies to Overcome Drug Resistance in Multiple Myeloma.Cancers (Basel). 2021 Apr 2;13(7):1686. doi: 10.3390/cancers13071686. Cancers (Basel). 2021. PMID: 33918370 Free PMC article. Review.
-
Wnt pathway activation and ABCB1 expression account for attenuation of proteasome inhibitor-mediated apoptosis in multidrug-resistant cancer cells.Cancer Biol Ther. 2015;16(1):149-59. doi: 10.4161/15384047.2014.987093. Cancer Biol Ther. 2015. PMID: 25590413 Free PMC article.
-
Drug and Solute Transporters in Mediating Resistance to Novel Therapeutics in Multiple Myeloma.ACS Pharmacol Transl Sci. 2021 Apr 15;4(3):1050-1065. doi: 10.1021/acsptsci.1c00074. eCollection 2021 Jun 11. ACS Pharmacol Transl Sci. 2021. PMID: 34151200 Free PMC article. Review.
-
Proteomics Analysis of Ovarian Cancer Cell Lines and Tissues Reveals Drug Resistance-associated Proteins.Cancer Genomics Proteomics. 2017 Jan 2;14(1):35-51. doi: 10.21873/cgp.20017. Cancer Genomics Proteomics. 2017. PMID: 28031236 Free PMC article.
-
Treatment with proteasome inhibitor bortezomib decreases organic anion transporting polypeptide (OATP) 1B3-mediated transport in a substrate-dependent manner.PLoS One. 2017 Nov 6;12(11):e0186924. doi: 10.1371/journal.pone.0186924. eCollection 2017. PLoS One. 2017. PMID: 29107984 Free PMC article.
References
-
- Richardson PG, Mitsiades C, Schlossman R, Munshi N, Anderson K. New drugs for myeloma. Oncologist. 2007;12:664–689. - PubMed
-
- O’Connor R. The pharmacology of cancer resistance. Anticancer Res. 2007;27:1267–1272. - PubMed
-
- Yang HH, Ma MH, Vescio RA, Berenson JR. Overcoming drug resistance in multiple myeloma: the emergence of therapeutic approaches to induce apoptosis. J Clin Oncol. 2003;21:4239–4247. - PubMed
-
- Grogan TM, Spier CM, Salmon SE, et al. P-glycoprotein expression in human plasma cell myeloma: correlation with prior chemotherapy. Blood. 1993;81:490–495. - PubMed
-
- Sonneveld P, Suciu S, Weijermans P, et al. Cyclosporin A combined with vincristine, doxorubicin and dexamethasone (VAD) compared with VAD alone in patients with advanced refractory multiple myeloma: an EORTC–HOVON randomized phase III study (06914) Br J Haematol. 2001;115:895–902. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

