Identification of salmonella pathogenicity island-2 type III secretion system effectors involved in intramacrophage replication of S. enterica serovar typhimurium: implications for rational vaccine design
- PMID: 23592259
- PMCID: PMC3634603
- DOI: 10.1128/mBio.00065-13
Identification of salmonella pathogenicity island-2 type III secretion system effectors involved in intramacrophage replication of S. enterica serovar typhimurium: implications for rational vaccine design
Abstract
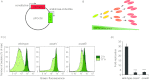
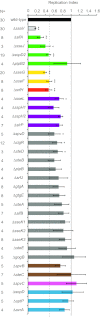
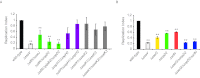
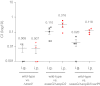
Salmonella enterica serovars cause severe diseases in humans, such as gastroenteritis and typhoid fever. The development of systemic disease is dependent on a type III secretion system (T3SS) encoded by Salmonella pathogenicity island-2 (SPI-2). Translocation of effector proteins across the Salmonella-containing vacuole, via the SPI-2 T3SS, enables bacterial replication within host cells, including macrophages. Here, we investigated the contribution of these effectors to intramacrophage replication of Salmonella enterica serovar Typhimurium using Fluorescence Dilution, a dual-fluorescence tool which allows direct measurement of bacterial replication. Of 32 strains, each carrying single mutations in genes encoding effectors, 10 (lacking sifA, sseJ, sopD2, sseG, sseF, srfH, sseL, spvD, cigR, or steD) were attenuated in replication in mouse bone marrow-derived macrophages. The replication profiles of strains combining deletions in effector genes were also investigated: a strain lacking the genes sseG, sopD2, and srfH showed an increased replication defect compared to single-mutation strains and was very similar to SPI-2 T3SS-deficient bacteria with respect to its replication defect. This strain was substantially attenuated in virulence in vivo and yet retained intracellular vacuole integrity and a functional SPI-2 T3SS. Moreover, this strain was capable of SPI-2 T3SS-mediated delivery of a model antigen for major histocompatibility complex (MHC) class I-dependent T-cell activation. This work establishes a basis for the use of a poly-effector mutant strain as an attenuated vaccine carrier for delivery of heterologous antigens directly into the cytoplasm of host cells.
Importance: Live attenuated strains of Salmonella enterica serotype Typhi have generated much interest in the search for improved vaccines against typhoid fever and as vaccine vectors for the delivery of heterologous antigens. A promising vaccine candidate is the ΔaroC ΔssaV S. Typhi strain, which owes its attenuation mainly to lack of a type III secretion system (SPI-2 T3SS). The SPI-2 T3SS is important for bacterial proliferation inside macrophages, but not all of the effectors involved in this process have been identified. Here, we show that 10 effectors of the related strain S. Typhimurium contribute to intracellular replication in macrophages. Moreover, we establish that a poly-effector mutant strain of S. Typhimurium can have a severe replication defect and maintain a functional SPI-2 T3SS, which can be exploited for delivery of heterologous antigens.
Figures

Similar articles
-
Salmonella enterica serovar Typhi uses two type 3 secretion systems to replicate in human macrophages and colonize humanized mice.mBio. 2023 Aug 31;14(4):e0113723. doi: 10.1128/mbio.01137-23. Epub 2023 Jun 21. mBio. 2023. PMID: 37341487 Free PMC article.
-
Salmonella Effectors SseF and SseG Interact with Mammalian Protein ACBD3 (GCP60) To Anchor Salmonella-Containing Vacuoles at the Golgi Network.mBio. 2016 Jul 12;7(4):e00474-16. doi: 10.1128/mBio.00474-16. mBio. 2016. PMID: 27406559 Free PMC article.
-
Evaluation of Salmonella enterica type III secretion system effector proteins as carriers for heterologous vaccine antigens.Infect Immun. 2012 Mar;80(3):1193-202. doi: 10.1128/IAI.06056-11. Epub 2012 Jan 17. Infect Immun. 2012. PMID: 22252866 Free PMC article.
-
Functions of the Salmonella pathogenicity island 2 (SPI-2) type III secretion system effectors.Microbiology (Reading). 2012 May;158(Pt 5):1147-1161. doi: 10.1099/mic.0.058115-0. Epub 2012 Mar 15. Microbiology (Reading). 2012. PMID: 22422755 Review.
-
Typhoidal Salmonella: Distinctive virulence factors and pathogenesis.Cell Microbiol. 2018 Sep;20(9):e12939. doi: 10.1111/cmi.12939. Epub 2018 Aug 9. Cell Microbiol. 2018. PMID: 30030897 Review.
Cited by
-
Dormant intracellular Salmonella enterica serovar Typhimurium discriminates among Salmonella pathogenicity island 2 effectors to persist inside fibroblasts.Infect Immun. 2014 Jan;82(1):221-32. doi: 10.1128/IAI.01304-13. Epub 2013 Oct 21. Infect Immun. 2014. PMID: 24144726 Free PMC article.
-
Quantitative proteomic screen identifies annexin A2 as a host target for Salmonella pathogenicity island-2 effectors SopD2 and PipB2.Sci Rep. 2021 Dec 8;11(1):23630. doi: 10.1038/s41598-021-02795-x. Sci Rep. 2021. PMID: 34880286 Free PMC article.
-
Genome sequencing and analysis of Salmonella enterica subsp. enterica serovar Stanley UPM 517: Insights on its virulence-associated elements and their potentials as vaccine candidates.PeerJ. 2019 Jun 28;7:e6948. doi: 10.7717/peerj.6948. eCollection 2019. PeerJ. 2019. PMID: 31293824 Free PMC article.
-
The Salmonella effector SseJ disrupts microtubule dynamics when ectopically expressed in normal rat kidney cells.PLoS One. 2017 Feb 24;12(2):e0172588. doi: 10.1371/journal.pone.0172588. eCollection 2017. PLoS One. 2017. PMID: 28235057 Free PMC article.
-
Salmonella Typhimurium infection inhibits macrophage IFNβ signaling in a TLR4-dependent manner.Infect Immun. 2024 Oct 15;92(10):e0009824. doi: 10.1128/iai.00098-24. Epub 2024 Sep 13. Infect Immun. 2024. PMID: 39269166 Free PMC article.
References
-
- van der Heijden J, Finlay BB. 2012. Type III effector-mediated processes in salmonella infection. Future Microbiol. 7:685–703 - PubMed
-
- Hensel M, Shea JE, Gleeson C, Jones MD, Dalton E, Holden DW. 1995. Simultaneous identification of bacterial virulence genes by negative selection. Science 269:400–403 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

