Impairment of FOS mRNA stabilization following translation arrest in granulocytes from myelodysplastic syndrome patients
- PMID: 23593403
- PMCID: PMC3625160
- DOI: 10.1371/journal.pone.0061107
Impairment of FOS mRNA stabilization following translation arrest in granulocytes from myelodysplastic syndrome patients
Expression of concern in
-
Expression of Concern: Impairment of FOS mRNA Stabilization Following Translation Arrest in Granulocytes from Myelodysplastic Syndrome Patients.PLoS One. 2023 Jan 11;18(1):e0278805. doi: 10.1371/journal.pone.0278805. eCollection 2023. PLoS One. 2023. PMID: 36630355 Free PMC article. No abstract available.
Abstract
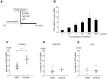
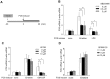
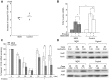
Although quantitative and qualitative granulocyte defects have been described in myelodysplastic syndromes (MDS), the underlying molecular basis of granulocyte dysfunction in MDS is largely unknown. We recently found that FOS mRNA elevation under translation-inhibiting stimuli was significantly smaller in granulocytes from MDS patients than in healthy individuals. The aim of this study is to clarify the cause of the impaired FOS induction in MDS. We first examined the mechanisms of FOS mRNA elevation using granulocytes from healthy donors cultured with the translation inhibitor emetine. Emetine increased both transcription and mRNA stability of FOS. p38 MAPK inhibition abolished the emetine-induced increase of FOS transcription but did not affect FOS mRNA stabilization. The binding of an AU-rich element (ARE)-binding protein HuR to FOS mRNA containing an ARE in 3'UTR was increased by emetine, and the knockdown of HuR reduced the FOS mRNA stabilizing effect of emetine. We next compared the emetine-induced transcription and mRNA stabilization of FOS between MDS patients and healthy controls. Increased rates of FOS transcription by emetine were similar in MDS and controls. In the absence of emetine, FOS mRNA decayed to nearly 17% of initial levels in 45 min in both groups. In the presence of emetine, however, 76.7±19.8% of FOS mRNA remained after 45 min in healthy controls, versus 37.9±25.5% in MDS (P<0.01). To our knowledge, this is the first report demonstrating attenuation of stress-induced FOS mRNA stabilization in MDS granulocytes.
Conflict of interest statement
Figures


Similar articles
-
Reduction of c-Fos via Overexpression of miR-34a Results in Enhancement of TNF- Production by LPS in Neutrophils from Myelodysplastic Syndrome Patients.PLoS One. 2016 Aug 11;11(8):e0158527. doi: 10.1371/journal.pone.0158527. eCollection 2016. PLoS One. 2016. PMID: 27513856 Free PMC article.
-
An interplay between the p38 MAPK pathway and AUBPs regulates c-fos mRNA stability during mitogenic stimulation.Biochem J. 2015 Apr 1;467(1):77-90. doi: 10.1042/BJ20141100. Biochem J. 2015. PMID: 25588078 Free PMC article.
-
Impairment of FOS mRNA induction by a translation inhibitor puromycin in granulocytes from myelodysplastic syndrome patients.Br J Haematol. 2011 Aug;154(4):525-7. doi: 10.1111/j.1365-2141.2010.08504.x. Epub 2011 Apr 11. Br J Haematol. 2011. PMID: 21480856 No abstract available.
-
The involvement of AU-rich element-binding proteins in p38 mitogen-activated protein kinase pathway-mediated mRNA stabilisation.Cell Signal. 2004 Oct;16(10):1113-21. doi: 10.1016/j.cellsig.2004.04.006. Cell Signal. 2004. PMID: 15240006 Review.
-
[Aberrant regulation of mRNA 3' untranslated region in cancers and inflammation].Med Sci (Paris). 2008 Mar;24(3):290-6. doi: 10.1051/medsci/2008243290. Med Sci (Paris). 2008. PMID: 18334178 Review. French.
Cited by
-
Rheum palmatum L. and Salvia miltiorrhiza Bge. Alleviates Acute Pancreatitis by Regulating Th17 Cell Differentiation: An Integrated Network Pharmacology Analysis, Molecular Dynamics Simulation and Experimental Validation.Chin J Integr Med. 2024 May;30(5):408-420. doi: 10.1007/s11655-023-3559-6. Epub 2023 Oct 20. Chin J Integr Med. 2024. PMID: 37861962
-
Expression of Concern: Impairment of FOS mRNA Stabilization Following Translation Arrest in Granulocytes from Myelodysplastic Syndrome Patients.PLoS One. 2023 Jan 11;18(1):e0278805. doi: 10.1371/journal.pone.0278805. eCollection 2023. PLoS One. 2023. PMID: 36630355 Free PMC article. No abstract available.
-
Reduction of c-Fos via Overexpression of miR-34a Results in Enhancement of TNF- Production by LPS in Neutrophils from Myelodysplastic Syndrome Patients.PLoS One. 2016 Aug 11;11(8):e0158527. doi: 10.1371/journal.pone.0158527. eCollection 2016. PLoS One. 2016. PMID: 27513856 Free PMC article.
-
Leptin in the Commissural Nucleus of the Tractus Solitarius (cNTS) and Anoxic Stimulus in the Carotid Body Chemoreceptors Increases cNTS Leptin Signaling Receptor and Brain Glucose Retention in Rats.Medicina (Kaunas). 2022 Apr 16;58(4):550. doi: 10.3390/medicina58040550. Medicina (Kaunas). 2022. PMID: 35454388 Free PMC article.
References
-
- Heaney ML, Golde DW (1999) Myelodysplasia. N Engl J Med 340: 1649–1660. - PubMed
-
- Shetty V, Hussaini S, Broady-Robinson L, Allampallam K, Mundle S, et al. (2000) Intramedullary apoptosis of hematopoietic cells in myelodysplastic syndrome patients can be massive: apoptotic cells recovered from high-density fraction of bone marrow aspirates. Blood 96: 1388–1392. - PubMed
-
- Shetty VT, Mundle SD, Raza A (2001) Pseudo Pelger-Huet anomaly in myelodysplastic syndrome: hyposegmented apoptotic neutrophil? Blood 98: 1273–1275. - PubMed
-
- Fianchi L, Leone G, Posteraro B, Sanguinetti M, Guidi F, et al. (2012) Impaired bactericidal and fungicidal activities of neutrophils in patients with myelodysplastic syndrome. Leuk Res 36: 331–333. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

