Polymyxins as novel and safe mucosal adjuvants to induce humoral immune responses in mice
- PMID: 23593492
- PMCID: PMC3623863
- DOI: 10.1371/journal.pone.0061643
Polymyxins as novel and safe mucosal adjuvants to induce humoral immune responses in mice
Abstract
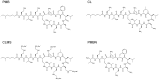
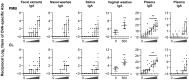
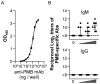
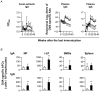
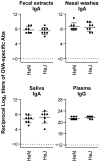
There is currently an urgent need to develop safe and effective adjuvants for enhancing vaccine-induced antigen-specific immune responses. We demonstrate here that intranasal immunization with clinically used polypeptide antibiotics, polymyxin B (PMB) and colistin (CL), along with ovalbumin (OVA), increases OVA-specific humoral immune responses in a dose-dependently manner at both mucosal and systemic compartments. Enhanced immunity by boosting was found to persist during 8 months of observation. Moreover, mice intranasally immunized with OVA plus various doses of PMB or CL showed neither inflammatory responses in the nasal cavity and olfactory bulbs nor renal damages, compared to those given OVA alone. These data suggest that polymyxins may serve as novel and safe mucosal adjuvants to induce humoral immune responses. The polymyxin adjuvanticity was found to be independent of endotoxins liberated by its bactericidal activity, as indicated by similar enhancing effects of PMB in lipopolysaccharide (LPS)-hyporesponsive and LPS-susceptible mice. However, despite the presence of preexisting anti-PMB antibodies, we observed no reduction in the adjuvant function of polymyxins when they were given intranasally. Furthermore, the titers of OVA-specific Abs in mice intranasally immunized with OVA plus PMB or CL were significantly higher than those in mice administered with polymyxin analogues, such as polymyxin B nonapeptide and colistin methanesulfonate. The levels of released β-hexosaminidase and histamine in mast cell culture supernatants stimulated by PMB or CL were also significantly higher than those stimulated by their analogues. These results suggest that both the hydrophobic carbon chain and hydrophilic cationic cyclic peptide contribute to the mucosal adjuvanticity of PMB and CL.
Conflict of interest statement
Figures
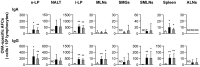

Similar articles
-
Mast cells partially contribute to mucosal adjuvanticity of surfactin in mice.Immun Inflamm Dis. 2018 Mar;6(1):117-127. doi: 10.1002/iid3.204. Epub 2017 Nov 3. Immun Inflamm Dis. 2018. PMID: 29105371 Free PMC article.
-
Suitability of Polymyxin B as a Mucosal Adjuvant for Intranasal Influenza and COVID-19 Vaccines.Vaccines (Basel). 2023 Nov 18;11(11):1727. doi: 10.3390/vaccines11111727. Vaccines (Basel). 2023. PMID: 38006059 Free PMC article.
-
Mucosal and systemic immune responses by intranasal immunization using archaeal lipid-adjuvanted vaccines.Vaccine. 2007 Dec 12;25(51):8622-36. doi: 10.1016/j.vaccine.2007.09.042. Epub 2007 Oct 5. Vaccine. 2007. PMID: 17959279
-
Pharmacokinetics/pharmacodynamics of colistin and polymyxin B: are we there yet?Int J Antimicrob Agents. 2016 Dec;48(6):592-597. doi: 10.1016/j.ijantimicag.2016.09.010. Epub 2016 Oct 18. Int J Antimicrob Agents. 2016. PMID: 27793510 Free PMC article. Review.
-
Population pharmacokinetics of polymyxin B: a systematic review.Ann Transl Med. 2022 Feb;10(4):231. doi: 10.21037/atm-22-236. Ann Transl Med. 2022. PMID: 35280373 Free PMC article. Review.
Cited by
-
MRGPR-mediated activation of local mast cells clears cutaneous bacterial infection and protects against reinfection.Sci Adv. 2019 Jan 2;5(1):eaav0216. doi: 10.1126/sciadv.aav0216. eCollection 2019 Jan. Sci Adv. 2019. PMID: 30613778 Free PMC article.
-
Mast cells partially contribute to mucosal adjuvanticity of surfactin in mice.Immun Inflamm Dis. 2018 Mar;6(1):117-127. doi: 10.1002/iid3.204. Epub 2017 Nov 3. Immun Inflamm Dis. 2018. PMID: 29105371 Free PMC article.
-
Mast cell activators as novel immune regulators.Curr Opin Pharmacol. 2018 Aug;41:89-95. doi: 10.1016/j.coph.2018.05.004. Epub 2018 May 26. Curr Opin Pharmacol. 2018. PMID: 29843056 Free PMC article. Review.
-
Roles and relevance of mast cells in infection and vaccination.J Biomed Res. 2016 Jul;30(4):253-63. doi: 10.7555/JBR.30.20150038. Epub 2015 Jun 29. J Biomed Res. 2016. PMID: 26565602 Free PMC article. Review.
-
Development of a broadly active influenza intranasal vaccine adjuvanted with self-assembled particles composed of mastoparan-7 and CpG.Front Immunol. 2023 Mar 24;14:1103765. doi: 10.3389/fimmu.2023.1103765. eCollection 2023. Front Immunol. 2023. PMID: 37033992 Free PMC article.
References
-
- Elson CO, Ealding W (1984) Generalized systemic and mucosal immunity in mice after mucosal stimulation with cholera toxin. J Immunol 132: 2736–2741. - PubMed
-
- Clements JD, Hartzog NM, Lyon FL (1988) Adjuvant activity of Escherichia coli heat-labile enterotoxin and effect on the induction of oral tolerance in mice to unrelated protein antigens. Vaccine 6: 269–277. - PubMed
-
- Mutsch M, Zhou W, Rhodes P, Bopp M, Chen RT, et al. (2004) Use of the inactivated intranasal influenza vaccine and the risk of Bell's palsy in Switzerland. N Engl J Med 350: 896–903. - PubMed
-
- McLachlan JB, Shelburne CP, Hart JP, Pizzo SV, Goyal R, et al. (2008) Mast cell activators: a new class of highly effective vaccine adjuvants. Nat Med 14: 536–541. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

