Identification of selective and potent inhibitors of fibroblast activation protein and prolyl oligopeptidase
- PMID: 23594271
- PMCID: PMC4059180
- DOI: 10.1021/jm400351a
Identification of selective and potent inhibitors of fibroblast activation protein and prolyl oligopeptidase
Abstract
Fibroblast activation protein (FAP) is a serine protease selectively expressed on reactive stromal fibroblasts of epithelial carcinomas. It is widely believed to play a role in tumor invasion and metastasis and therefore to represent a potential new drug target for cancer. Investigation into its biological function, however, has been hampered by the current unavailability of selective inhibitors. The challenge has been in identifying inhibitors that are selective for FAP over both the dipeptidyl peptidases (DPPs), with which it shares exopeptidase specificity, and prolyl oligopeptidase (PREP), with which it shares endopeptidase specificity. Here, we report the first potent FAP inhibitor with selectivity over both the DPPs and PREP, N-(pyridine-4-carbonyl)-d-Ala-boroPro (ARI-3099, 6). We also report a similarly potent and selective PREP inhibitor, N-(pyridine-3-carbonyl)-Val-boroPro (ARI-3531, 22). Both are boronic acid based inhibitors, demonstrating that high selectivity can be achieved using this electrophile. The inhibitors are stable, easy to synthesize, and should prove to be useful in helping to elucidate the biological functions of these two unique and interesting enzymes, as well as their potential as drug targets.
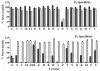
Figures




References
-
- Rettig WJ, Garin-Chesa P, Healey JH, Su SL, Ozer HL, Schwab M, Albino AP, Old LJ. Regulation and heteromeric structure of the fibroblast activation protein in normal and transformed cells of mesenchymal and neuroectodermal origin. Cancer Res. 1993;53:3327–3335. - PubMed
-
- Rettig WJ, Su SL, Fortunato SR, Scanlan MJ, Raj BK, Garin-Chesa P, Healey JH, Old LJ. Fibroblast activation protein: purification, epitope mapping and induction by growth factors. Int. J. Cancer. 1994;58:385–392. - PubMed
-
- Park JE, Lenter MC, Zimmermann RN, Garin-Chesa P, Old LJ, Rettig WJ. Fibroblast activation protein, a dual specificity serine protease expressed in reactive human tumor stromal fibroblasts. J. Biol. Chem. 1999;274:36505–36512. - PubMed
-
- Connolly BA, Sanford DG, Chiluwal AK, Healey SE, Peters DE, Dimare MT, Wu W, Liu Y, Maw H, Zhou Y, Li Y, Jin Z, Sudmeier JL, Lai JH, Bachovchin WW. Dipeptide boronic acid inhibitors of dipeptidyl peptidase IV: determinants of potency and in vivo efficacy and safety. J. Med. Chem. 2008;51:6005–6013. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
Molecular Biology Databases
Miscellaneous

