Endothelial mineralocorticoid receptor activation mediates endothelial dysfunction in diet-induced obesity
- PMID: 23594590
- PMCID: PMC3844149
- DOI: 10.1093/eurheartj/eht095
Endothelial mineralocorticoid receptor activation mediates endothelial dysfunction in diet-induced obesity
Abstract
Aims: Aldosterone plays a crucial role in cardiovascular disease. 'Systemic' inhibition of its mineralocorticoid receptor (MR) decreases atherosclerosis by reducing inflammation and oxidative stress. Obesity, an important cardiovascular risk factor, is an inflammatory disease associated with increased plasma aldosterone levels. We have investigated the role of the 'endothelial' MR in obesity-induced endothelial dysfunction, the earliest stage in atherogenesis.
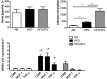
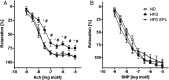
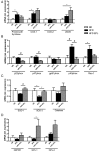
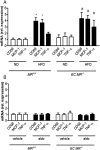
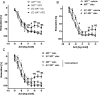
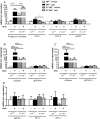
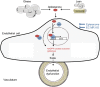
Methods and results: C57BL/6 mice were exposed to a normal chow diet (ND) or a high-fat diet (HFD) alone or in combination with the MR antagonist eplerenone (200 mg/kg/day) for 14 weeks. Diet-induced obesity impaired endothelium-dependent relaxation in response to acetylcholine, whereas eplerenone treatment of obese mice prevented this. Expression analyses in aortic endothelial cells isolated from these mice revealed that eplerenone attenuated expression of pro-oxidative NADPH oxidase (subunits p22phox, p40phox) and increased expression of antioxidative genes (glutathione peroxidase-1, superoxide dismutase-1 and -3) in obesity. Eplerenone did not affect obesity-induced upregulation of cyclooxygenase (COX)-1 or prostacyclin synthase. Endothelial-specific MR deletion prevented endothelial dysfunction in obese (exhibiting high 'endogenous' aldosterone) and in 'exogenous' aldosterone-infused lean mice. Pre-incubation of aortic rings from aldosterone-treated animals with the COX-inhibitor indomethacin restored endothelial function. Exogenous aldosterone administration induced endothelial expression of p22phox in the presence, but not in the absence of the endothelial MR.
Conclusion: Obesity-induced endothelial dysfunction depends on the 'endothelial' MR and is mediated by an imbalance of oxidative stress-modulating mechanisms. Therefore, MR antagonists may represent an attractive therapeutic strategy in the increasing population of obese patients to decrease vascular dysfunction and subsequent atherosclerotic complications.
Keywords: Aldosterone; Endothelial; Mineralocorticoid receptor; Obesity.
Figures
Comment in
-
Mineralocorticoid receptor throughout the vessel: a key to vascular dysfunction in obesity.Eur Heart J. 2013 Dec;34(45):3475-7. doi: 10.1093/eurheartj/eht158. Epub 2013 May 10. Eur Heart J. 2013. PMID: 23666249 Free PMC article. No abstract available.
References
-
- James PT, Rigby N, Leach R. The obesity epidemic, metabolic syndrome and future prevention strategies. Eur J Cardiovasc Prev Rehab. 2004;355:763–778. - PubMed
-
- Spiegelman BM, Flier JS. Obesity and the regulation of energy balance. Cell. 2001;104:531–543. - PubMed
-
- Ahima RS. Adipose tissue as an endocrin organ. Obesity. 2006;14:242S–249S. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

