Glycopolymer probes of signal transduction
- PMID: 23595539
- PMCID: PMC3808984
- DOI: 10.1039/c3cs60097a
Glycopolymer probes of signal transduction
Abstract
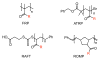
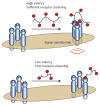
Glycans are key participants in biological processes ranging from reproduction to cellular communication to infection. Revealing glycan roles and the underlying molecular mechanisms by which glycans manifest their function requires access to glycan derivatives that vary systematically. To this end, glycopolymers (polymers bearing pendant carbohydrates) have emerged as valuable glycan analogs. Because glycopolymers can readily be synthesized, their overall shape can be varied, and they can be altered systematically to dissect the structural features that underpin their activities. This review provides examples in which glycopolymers have been used to effect carbohydrate-mediated signal transduction. Our objective is to illustrate how these powerful tools can reveal the molecular mechanisms that underlie carbohydrate-mediated signal transduction.
Figures
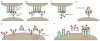

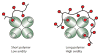


Similar articles
-
Noncovalent cell surface engineering: incorporation of bioactive synthetic glycopolymers into cellular membranes.J Am Chem Soc. 2008 May 7;130(18):5947-53. doi: 10.1021/ja710644g. Epub 2008 Apr 11. J Am Chem Soc. 2008. PMID: 18402449 Free PMC article.
-
Nanoscale materials for probing the biological functions of the glycocalyx.Glycobiology. 2016 Aug;26(8):797-803. doi: 10.1093/glycob/cww022. Epub 2016 Feb 24. Glycobiology. 2016. PMID: 26916883 Free PMC article. Review.
-
Metabolic Chemical Reporters of Glycans Exhibit Cell-Type-Selective Metabolism and Glycoprotein Labeling.Chembiochem. 2017 Jul 4;18(13):1177-1182. doi: 10.1002/cbic.201700020. Epub 2017 Mar 28. Chembiochem. 2017. PMID: 28231413 Free PMC article.
-
Chemoenzymatic modular assembly of O-GalNAc glycans for functional glycomics.Nat Commun. 2021 Jun 11;12(1):3573. doi: 10.1038/s41467-021-23428-x. Nat Commun. 2021. PMID: 34117223 Free PMC article.
-
Tools for Studying Glycans: Recent Advances in Chemoenzymatic Glycan Labeling.ACS Chem Biol. 2017 Mar 17;12(3):611-621. doi: 10.1021/acschembio.6b01089. Epub 2017 Feb 13. ACS Chem Biol. 2017. PMID: 28301937 Free PMC article. Review.
Cited by
-
Structure and Dynamics of N-Glycosylated Human Ribonuclease 1.Biochemistry. 2020 Sep 1;59(34):3148-3156. doi: 10.1021/acs.biochem.0c00191. Epub 2020 Jun 30. Biochemistry. 2020. PMID: 32544330 Free PMC article.
-
Multivalent Antigens for Promoting B and T Cell Activation.ACS Chem Biol. 2015 Aug 21;10(8):1817-24. doi: 10.1021/acschembio.5b00239. Epub 2015 Jun 2. ACS Chem Biol. 2015. PMID: 25970017 Free PMC article.
-
Chemoselective, Postpolymerization Modification of Bioactive, Degradable Polymers.Biomacromolecules. 2019 Feb 11;20(2):1018-1027. doi: 10.1021/acs.biomac.8b01631. Epub 2019 Jan 22. Biomacromolecules. 2019. PMID: 30608163 Free PMC article.
-
Multicomponent reaction derived small di- and tri-carbohydrate-based glycomimetics as tools for probing lectin specificity.Glycoconj J. 2022 Oct;39(5):587-597. doi: 10.1007/s10719-022-10079-3. Epub 2022 Aug 24. Glycoconj J. 2022. PMID: 36001188
-
Stereoelectronic Effects Impact Glycan Recognition.J Am Chem Soc. 2020 Feb 5;142(5):2386-2395. doi: 10.1021/jacs.9b11699. Epub 2020 Jan 24. J Am Chem Soc. 2020. PMID: 31930911 Free PMC article.
References
-
- Ito S. Philos Trans R Soc Lond B Biol Sci. 1974;268:55–66. - PubMed
-
- Costerton JW, Irvin RT, Cheng KJ. Annu Rev Microbiol. 1981;35:299–324. - PubMed
-
- Schröter S, Osterhoff C, McArdle W, Ivell R. Hum Reprod Update. 1999;5:302–313. - PubMed
-
- Pang PC, Chiu PCN, Lee CL, Chang LY, Panico M, Morris HR, Haslam SM, Khoo KH, Clark GF, Yeung WSB, Dell A. Science. 2011;333:1761–1764. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

