Connexin mutants and cataracts
- PMID: 23596416
- PMCID: PMC3625720
- DOI: 10.3389/fphar.2013.00043
Connexin mutants and cataracts
Abstract
The lens is a multicellular, but avascular tissue that must stay transparent to allow normal transmission of light and focusing of it on the retina. Damage to lens cells and/or proteins can cause cataracts, opacities that disrupt these processes. The normal survival of the lens is facilitated by an extensive network of gap junctions formed predominantly of connexin46 and connexin50. Mutations of the genes that encode these connexins (GJA3 and GJA8) have been identified and linked to inheritance of cataracts in human families and mouse lines. In vitro expression studies of several of these mutants have shown that they exhibit abnormalities that may lead to disease. Many of the mutants reduce or modify intercellular communication due to channel alterations (including loss of function or altered gating) or due to impaired cellular trafficking which reduces the number of gap junction channels within the plasma membrane. However, the abnormalities detected in studies of other mutants suggest that they cause cataracts through other mechanisms including gain of hemichannel function (leading to cell injury and death) and formation of cytoplasmic accumulations (that may act as light scattering particles). These observations and the anticipated results of ongoing studies should elucidate the mechanisms of cataract development due to mutations of lens connexins and abnormalities of other lens proteins. They may also contribute to our understanding of the mechanisms of disease due to connexin mutations in other tissues.
Keywords: cataract; connexin46; connexin50; gap junction; lens.
Figures


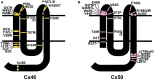
 ) and frame shift (
) and frame shift ( ) mutations identified in members of families with inherited cataracts. While included in Table 2, Cx50I247M has not been included in the Cx50 diagram, since it may actually be a polymorphism.
) mutations identified in members of families with inherited cataracts. While included in Table 2, Cx50I247M has not been included in the Cx50 diagram, since it may actually be a polymorphism.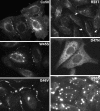
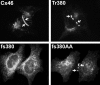

References
-
- Addison P. K., Berry V., Holden K. R., Espinal D., Rivera B., Su H., et al. (2006). A novel mutation in the connexin 46 gene (GJA3) causes autosomal dominant zonular pulverulent cataract in a Hispanic family. Mol. Vis. 12, 791–795 - PubMed
-
- Arora A., Minogue P. J., Liu X., Reddy M. A., Ainsworth J. R., Bhattacharya S. S., et al. (2006). A novel GJA8 mutation is associated with autosomal dominant lamellar pulverulent cataract: further evidence for gap junction dysfunction in human cataract. J. Med. Genet. 43, e2.10.1136/jmg.2005.034108 - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

