High frequency dynamic nuclear polarization
- PMID: 23597038
- PMCID: PMC3778063
- DOI: 10.1021/ar300348n
High frequency dynamic nuclear polarization
Abstract
During the three decades 1980-2010, magic angle spinning (MAS) NMR developed into the method of choice to examine many chemical, physical, and biological problems. In particular, a variety of dipolar recoupling methods to measure distances and torsion angles can now constrain molecular structures to high resolution. However, applications are often limited by the low sensitivity of the experiments, due in large part to the necessity of observing spectra of low-γ nuclei such as the I = 1/2 species (13)C or (15)N. The difficulty is still greater when quadrupolar nuclei, such as (17)O or (27)Al, are involved. This problem has stimulated efforts to increase the sensitivity of MAS experiments. A particularly powerful approach is dynamic nuclear polarization (DNP) which takes advantage of the higher equilibrium polarization of electrons (which conventionally manifests in the great sensitivity advantage of EPR over NMR). In DNP, the sample is doped with a stable paramagnetic polarizing agent and irradiated with microwaves to transfer the high polarization in the electron spin reservoir to the nuclei of interest. The idea was first explored by Overhauser and Slichter in 1953. However, these experiments were carried out on static samples, at magnetic fields that are low by current standards. To be implemented in contemporary MAS NMR experiments, DNP requires microwave sources operating in the subterahertz regime, roughly 150-660 GHz, and cryogenic MAS probes. In addition, improvements were required in the polarizing agents, because the high concentrations of conventional radicals that are required to produce significant enhancements compromise spectral resolution. In the last two decades, scientific and technical advances have addressed these problems and brought DNP to the point where it is achieving wide applicability. These advances include the development of high frequency gyrotron microwave sources operating in the subterahertz frequency range. In addition, low temperature MAS probes were developed that permit in situ microwave irradiation of the samples. And, finally, biradical polarizing agents were developed that increased the efficiency of DNP experiments by factors of ∼4 at considerably lower paramagnet concentrations. Collectively, these developments have made it possible to apply DNP on a routine basis to a number of different scientific endeavors, most prominently in the biological and material sciences. This Account reviews these developments, including the primary mechanisms used to transfer polarization in high frequency DNP, and the current choice of microwave sources and biradical polarizing agents. In addition, we illustrate the utility of the technique with a description of applications to membrane and amyloid proteins that emphasizes the unique structural information that is available in these two cases.
Figures


 , respectively. The mixing of states is proportional to a constant q, which is inversely proportional to B0. Therefore, the enhancement in the Solid Effect DNP scales as
. (bottom) A plot of the enhancement from SA-BDPA as a function of magnetic field (1H frequency) showing the positive and negative enhancements. ωNMR and ωEPR are the NMR and EPR frequencies and ωe ±ωn are the sum and difference of the EPR and NMR frequencies.
, respectively. The mixing of states is proportional to a constant q, which is inversely proportional to B0. Therefore, the enhancement in the Solid Effect DNP scales as
. (bottom) A plot of the enhancement from SA-BDPA as a function of magnetic field (1H frequency) showing the positive and negative enhancements. ωNMR and ωEPR are the NMR and EPR frequencies and ωe ±ωn are the sum and difference of the EPR and NMR frequencies.
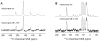



References
-
- Griffin RG. Dipolar recoupling in MAS spectra of biological solids. Nature Structural Biology. 1998;5:508–512. - PubMed
-
- Rienstra CM, Hohwy M, Mueller LJ, Jaroniec CP, Reif B, Griffin RG. Determination of multiple torsion-angle constraints in U-13C,15N-labeled peptides: 3D 1H-15N-13C-1H dipolar chemical shift spectroscopy in rotating solids. J Am Chem Soc. 2002;124:11908–11922. - PubMed
-
- Thompson LK, McDermott AE, Raap J, van der Wielen CM, Lugtenburg J, Herzfeld J, Griffin RG. Rotational Resonance NMR Study of the Active Site Structure in Bacteriorhodopsin: COnformation of the Schiff Base Linkage. Biochemistry. 1992;31:7931. - PubMed
-
- Griffiths JM, Lakshmi KV, Bennett AE, Raap J, Vanderwielen CM, Lugtenburg J, Herzfeld J, Griffin RG. Dipolar Correlation NMR-Spectroscopy of a Membrane-Protein. J Am Chem Soc. 1994;116:10178–10181.
Publication types
MeSH terms
Substances
Grants and funding
- EB002026/EB/NIBIB NIH HHS/United States
- R01 EB001965/EB/NIBIB NIH HHS/United States
- P41 EB002026/EB/NIBIB NIH HHS/United States
- R01 EB002804/EB/NIBIB NIH HHS/United States
- GM095843/GM/NIGMS NIH HHS/United States
- R01 EB001035/EB/NIBIB NIH HHS/United States
- R01 EB004866/EB/NIBIB NIH HHS/United States
- R01 EB001960/EB/NIBIB NIH HHS/United States
- EB001035/EB/NIBIB NIH HHS/United States
- R01 EB003151/EB/NIBIB NIH HHS/United States
- EB002804/EB/NIBIB NIH HHS/United States
- EB003151/EB/NIBIB NIH HHS/United States
- R01 GM095843/GM/NIGMS NIH HHS/United States
- EB001960/EB/NIBIB NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources

