Loss of TSC2 confers resistance to ceramide and nutrient deprivation
- PMID: 23604129
- PMCID: PMC3858574
- DOI: 10.1038/onc.2013.139
Loss of TSC2 confers resistance to ceramide and nutrient deprivation
Abstract
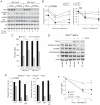
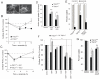
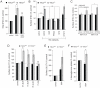
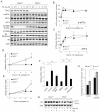
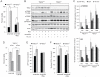
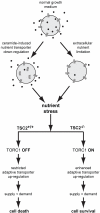
Nutrient stress that produces quiescence and catabolism in normal cells is lethal to cancer cells, because oncogenic mutations constitutively drive anabolism. One driver of biosynthesis in cancer cells is the mammalian target of rapamycin complex 1 (mTORC1) signaling complex. Activating mTORC1 by deleting its negative regulator tuberous sclerosis complex 2 (TSC2) leads to hypersensitivity to glucose deprivation. We have previously shown that ceramide kills cells in part by triggering nutrient transporter loss and restricting access to extracellular amino acids and glucose, suggesting that TSC2-deficient cells would be hypersensitive to ceramide. However, murine embryonic fibroblasts (MEFs) lacking TSC2 were highly resistant to ceramide-induced death. Consistent with the observation that ceramide limits access to both amino acids and glucose, TSC2(-/-) MEFs also had a survival advantage when extracellular amino acids and glucose were both reduced. As TSC2(-/-) MEFs were resistant to nutrient stress despite sustained mTORC1 activity, we assessed whether mTORC1 signaling might be beneficial under these conditions. In low amino acid and glucose medium, and following ceramide-induced nutrient transporter loss, elevated mTORC1 activity significantly enhanced the adaptive upregulation of new transporter proteins for amino acids and glucose. Strikingly, the introduction of oncogenic Ras abrogated the survival advantage of TSC2(-/-) MEFs upon ceramide treatment most likely by increasing nutrient demand. These results suggest that, in the absence of oncogene-driven biosynthetic demand, mTORC1-dependent translation facilitates the adaptive cellular response to nutrient stress.
Figures


References
-
- Dazert E, Hall MN. mTOR signaling in disease. Curr Opin Cell Biol. 2011;23:744–55. - PubMed
-
- Yecies JL, Manning BD. mTOR links oncogenic signaling to tumor cell metabolism. Journal of molecular medicine. 2011;89:221–8. - PubMed
-
- Curatolo P, Bombardieri R, Jozwiak S. Tuberous sclerosis. Lancet. 2008;372:657–68. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

