Role of mTOR inhibition in preventing resistance and restoring sensitivity to hormone-targeted and HER2-targeted therapies in breast cancer
- PMID: 23604238
- PMCID: PMC3774138
Role of mTOR inhibition in preventing resistance and restoring sensitivity to hormone-targeted and HER2-targeted therapies in breast cancer
Abstract
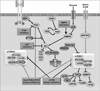
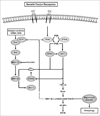
Even with hormone-targeted and human epidermal growth factor receptor 2 (HER2)-targeted anticancer agents, intrinsic resistance or acquired resistance are common occurrences in estrogen receptor-positive and HER2-positive breast cancers, respectively. Potential mechanisms for resistance to targeted agents include steric inhibition imposed by other cellular elements, molecular changes in the target receptor, alterations in the regulation of downstream signaling components, compensatory cross-talk with other signaling pathways, and pharmacogenetic alterations in the host. Evidence suggests that both hormone receptor-positive tumors and HER2-overexpressing tumors use the phosphoinositide 3-kinase/Akt/ mammalian target of rapamycin (mTOR) pathway to escape control of antihormone and anti-HER2 therapies. The combination of mTOR inhibitors with hormone-targeted or HER2-targeted therapies appears to be a promising strategy for overcoming resistant disease and preventing the development of resistance.
Figures
Similar articles
-
Potential of overcoming resistance to HER2-targeted therapies through the PI3K/Akt/mTOR pathway.Breast. 2015 Oct;24(5):548-55. doi: 10.1016/j.breast.2015.06.002. Epub 2015 Jul 15. Breast. 2015. PMID: 26187798 Review.
-
Overcoming resistance and restoring sensitivity to HER2-targeted therapies in breast cancer.Ann Oncol. 2012 Dec;23(12):3007-3016. doi: 10.1093/annonc/mds200. Epub 2012 Aug 2. Ann Oncol. 2012. PMID: 22865781 Free PMC article. Review.
-
The emergence of targeted drugs in breast cancer to prevent resistance to endocrine treatment and chemotherapy.Expert Opin Pharmacother. 2014 Apr;15(5):681-700. doi: 10.1517/14656566.2014.885952. Epub 2014 Feb 28. Expert Opin Pharmacother. 2014. PMID: 24579888 Review.
-
mTOR inhibitors in advanced breast cancer: ready for prime time?Cancer Treat Rev. 2013 Nov;39(7):742-52. doi: 10.1016/j.ctrv.2013.02.005. Epub 2013 Apr 1. Cancer Treat Rev. 2013. PMID: 23557794 Review.
-
Targeting PI3K/mTOR overcomes resistance to HER2-targeted therapy independent of feedback activation of AKT.Clin Cancer Res. 2014 Jul 1;20(13):3507-20. doi: 10.1158/1078-0432.CCR-13-2769. Epub 2014 May 30. Clin Cancer Res. 2014. PMID: 24879796
Cited by
-
RAD001 can reverse drug resistance of SGC7901/DDP cells.Tumour Biol. 2014 Sep;35(9):9171-7. doi: 10.1007/s13277-014-1719-1. Epub 2014 Jun 12. Tumour Biol. 2014. PMID: 24920069
-
Novel Strategies to Improve the Endocrine Therapy of Breast Cancer.Oncol Rev. 2017 May 16;11(1):323. doi: 10.4081/oncol.2017.323. eCollection 2017 Mar 3. Oncol Rev. 2017. PMID: 28584571 Free PMC article. Review.
-
Adipocyte/Tumor cell crosstalk via IGF-1/TXNIP axis promotes malignancy and endocrine resistance in breast cancer.Cell Commun Signal. 2025 Jun 3;23(1):262. doi: 10.1186/s12964-025-02262-4. Cell Commun Signal. 2025. PMID: 40462107 Free PMC article.
-
An overview of resistance to Human epidermal growth factor receptor 2 (Her2) targeted therapies in breast cancer.Cancer Drug Resist. 2022 Jun 1;5(2):472-486. doi: 10.20517/cdr.2022.09. eCollection 2022. Cancer Drug Resist. 2022. PMID: 35800378 Free PMC article. Review.
-
Inhibition of Aerobic Glycolysis Represses Akt/mTOR/HIF-1α Axis and Restores Tamoxifen Sensitivity in Antiestrogen-Resistant Breast Cancer Cells.PLoS One. 2015 Jul 9;10(7):e0132285. doi: 10.1371/journal.pone.0132285. eCollection 2015. PLoS One. 2015. PMID: 26158266 Free PMC article.
References
-
- Perou CM, Sorlie T, Eisen MB, et al. Molecular portraits of human breast tumours. Nature. 2000;406:747–752. - PubMed
-
- Hurvitz SA, Pietras RJ. Rational management of endocrine resistance in breast cancer: a comprehensive review of estrogen receptor biology, treatment options, and future directions. Cancer. 2008;113:2385–2397. - PubMed
-
- Arteaga CL, Sliwkowski MX, Osborne CK, Perez EA, Puglisi F, Gianni L. Treatment of HER2-positive breast cancer: current status and future perspectives. Nat Rev Clin Oncol. 2012;9:16–32. - PubMed
-
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Breast Cancer Screening and Diagnosis, V1.2011. Fort Washington, PA: NCCN; 2011. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
