Polypharmacy and risk of antiretroviral drug interactions among the aging HIV-infected population
- PMID: 23605401
- PMCID: PMC3785656
- DOI: 10.1007/s11606-013-2449-6
Polypharmacy and risk of antiretroviral drug interactions among the aging HIV-infected population
Abstract
Background: Among aging HIV-infected adults, polypharmacy and its consequences have not been well-described.
Objective: To characterize the extent of polypharmacy and the risk of antiretroviral (ARV) drug interactions among persons of different ages.
Design and participants: Cross-sectional analysis among patients within the HIV Outpatient Study (HOPS) cohort who were prescribed ARVs during 2006-2010.
Main measures: We used the University of Liverpool HIV drug interactions database to identify ARV/non-ARV interactions with potential for clinical significance.
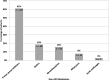
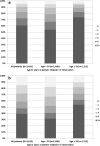
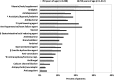
Key results: Of 3,810 patients analyzed (median age 46 years, 34 % ≥ 50 years old) at midpoint of observation, 1,494 (39 %) patients were prescribed ≥ 5 non-ARV medications: 706 (54 %) of 1,312 patients ≥ 50 years old compared with 788 (32 %) of 2,498 patients < 50 years. During the five-year period, the number of patients who were prescribed at least one ARV/non-ARV combination that was contraindicated or had moderate or high evidence of interaction was 267 (7 %) and 1,267 (33 %), respectively. Variables independently associated with having been prescribed a contraindicated ARV/non-ARV combination included older age (adjusted odds ratio [aOR] per 10 years of age 1.17, 95 % CI 1.01-1.35), anxiety (aOR 1.78, 95 % CI 1.32-2.40), dyslipidemia (aOR 1.96, 95 % CI 1.28-2.99), higher daily non-ARV medication burden (aOR 1.13, 95 % CI 1.10-1.17), and having been prescribed a protease inhibitor (aOR 2.10, 95 % CI 1.59-2.76). Compared with patients < 50 years, older patients were more likely to have been prescribed an ARV/non-ARV combination that was contraindicated (unadjusted OR 1.44, 95 % CI 1.14-1.82), or had moderate or high evidence of interaction (unadjusted OR 1.29, 95 % CI 1.15-1.44).
Conclusions: A substantial percentage of patients were prescribed at least one ARV/non-ARV combination that was contraindicated or had potential for a clinically significant interaction. As HIV-infected patients age and experience multiple comorbidities, systematic reviews of current medications by providers may reduce risk of such exposures.
Figures
References
-
- Crum NF, Riffenburgh RH, Wegner S, et al. Comparisons of causes of death and mortality rates among HIV-infected persons: analysis of the pre-, early, and late HAART (highly active antiretroviral therapy) eras. J Acquir Immune Defic Syndr. 2006;41(2):194–200. doi: 10.1097/01.qai.0000179459.31562.16. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

